Mix & Match! Buy 5 Products, Get 1 Free! Use code B5G1. Get Started!
Product: GEM 21S®

BIOBRIEF
Vertical Bone Augmentation with Bone Plate Technique and Soft Tissue Phenotype Modification


THE SITUATION
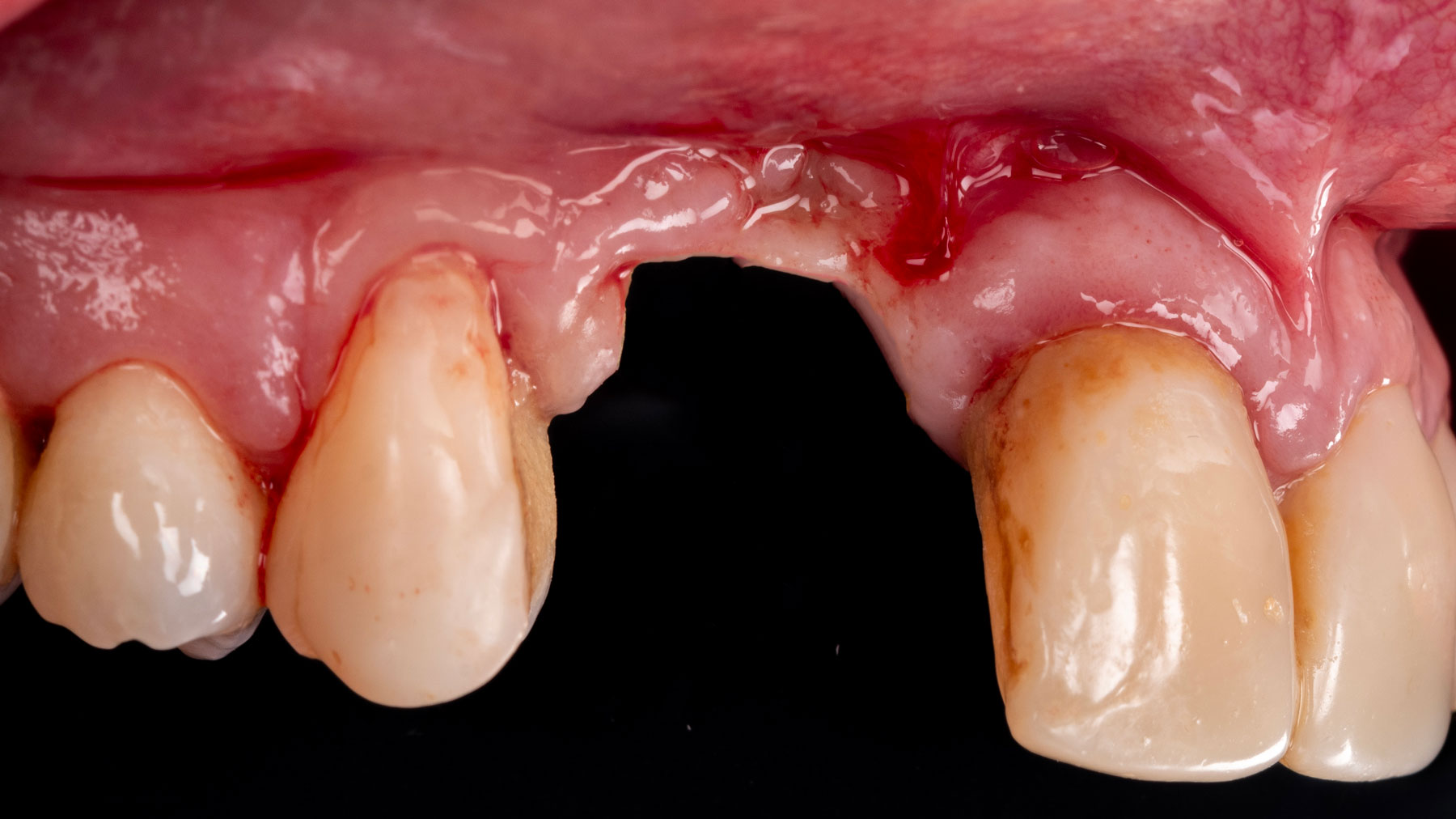
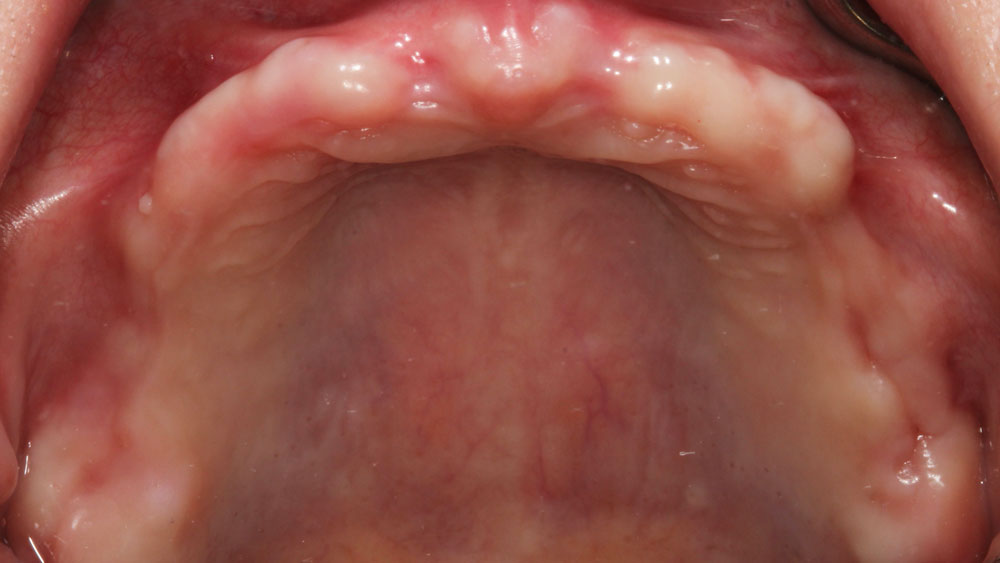
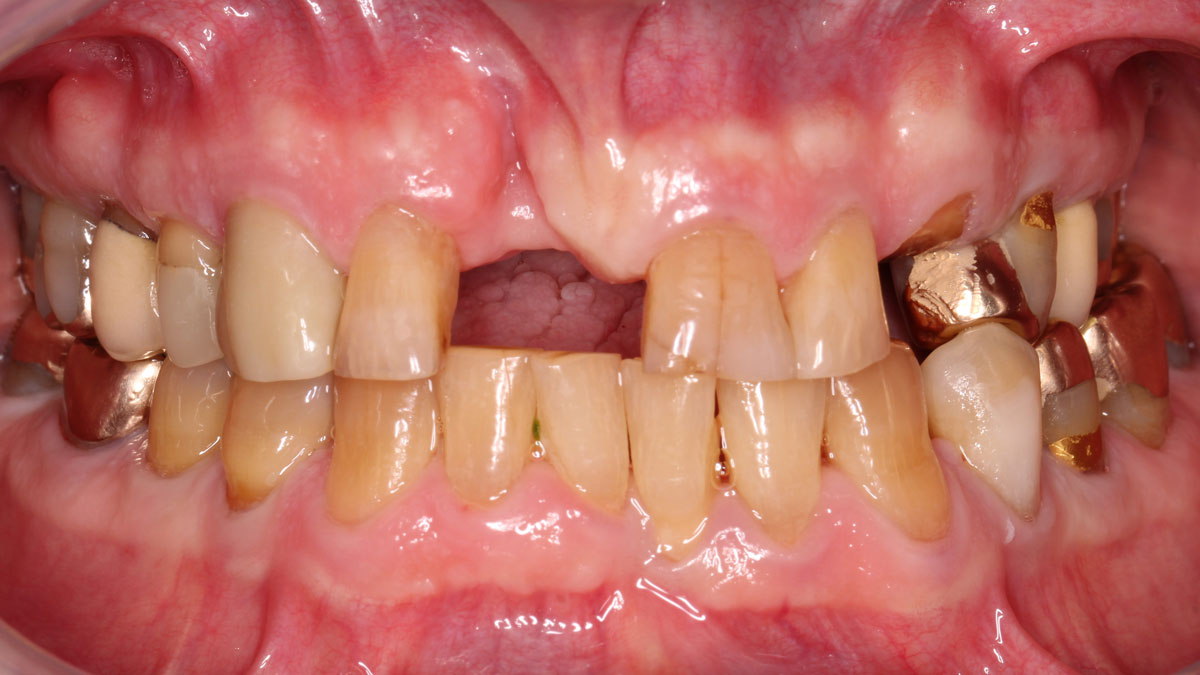
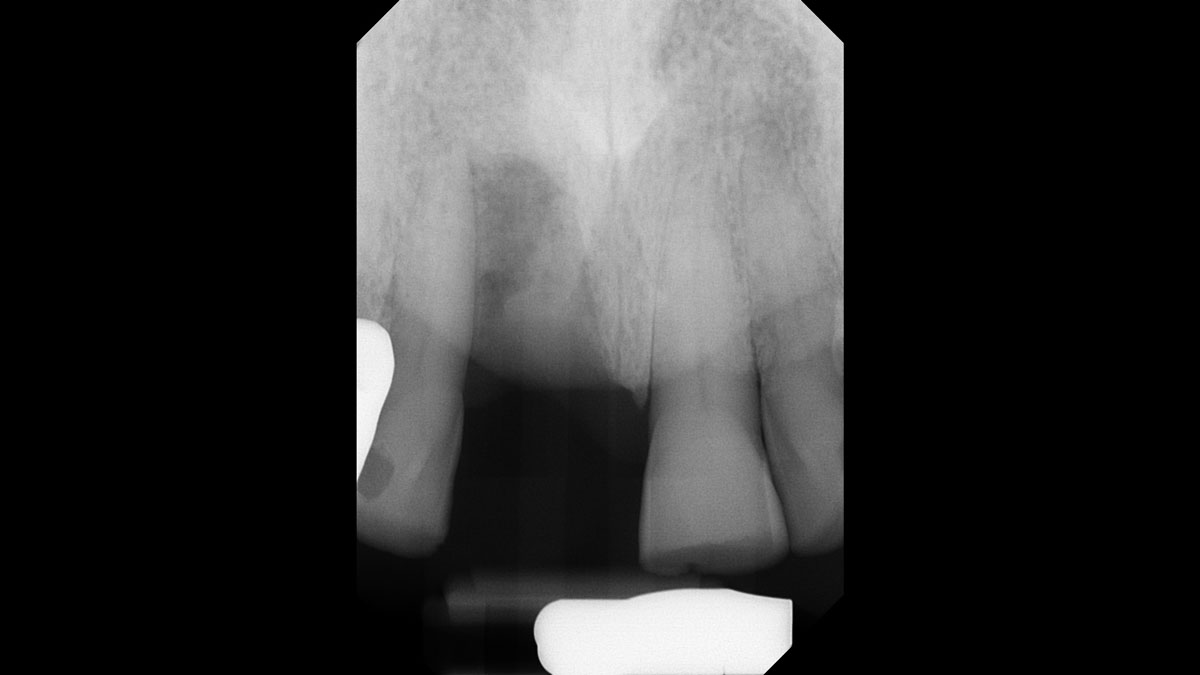
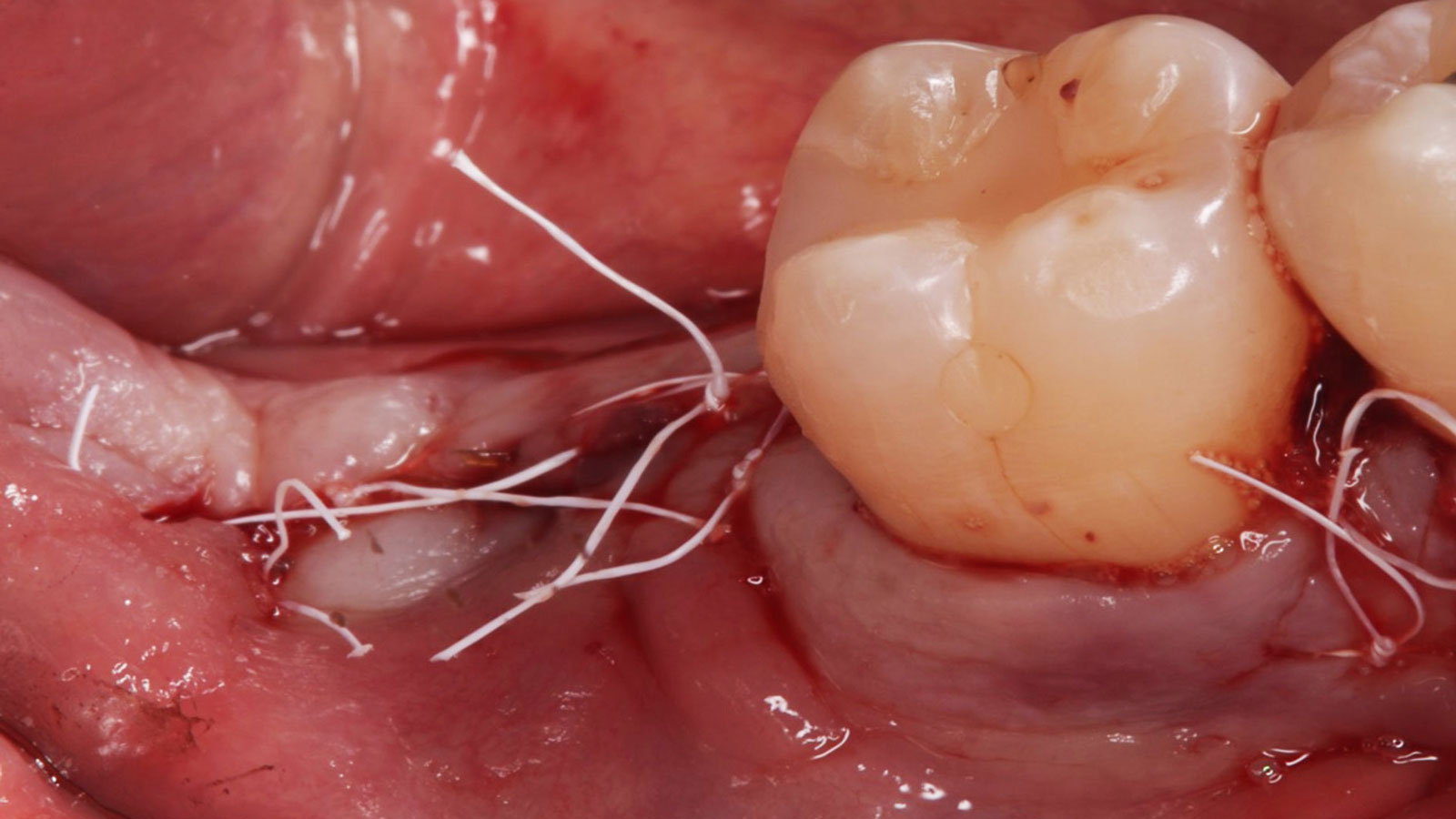
A patient presented with a vertical bone defect in the maxillary anterior region and expressed high esthetic demands while explicitly wishing to avoid autogenous grafting. This required a regenerative strategy that could reliably achieve vertical augmentation, maintain space, and deliver a stable peri-implant soft tissue phenotype—all without harvesting autogenous blocks or connective tissue.
THE RISK PROFILE
| Low Risk | Medium Risk | High Risk | |
|---|---|---|---|
| Patient’s esthetic requirements | Low | Medium | High |
| Height of the smile line | Low | Medium | High |
| Gingival biotype | Thick – “low scalloped” | Medium – “medium scalloped” | Thin – “high scalloped” |
| Infection at implant sight | None | Chronic | Acute |
| Bone height at adjacent tooth | ≤ 5 mm from contact point | 5.5 – 6.5 mm from contact point | ≥ 7 mm from contact point |
| Restorative status of adjacent tooth | Intact | Restored (#8 has been restored) | |
| Width of tooth gap | 1 tooth (≥ 7 mm) | 1 tooth (≤ 7 mm) | 2 teeth or more |
| Soft-tissue anatomy | Intact | Compromised | |
| Bone anatomy of the alveolar ridge | No defect | Horizontal defect | Vertical defect |
THE APPROACH
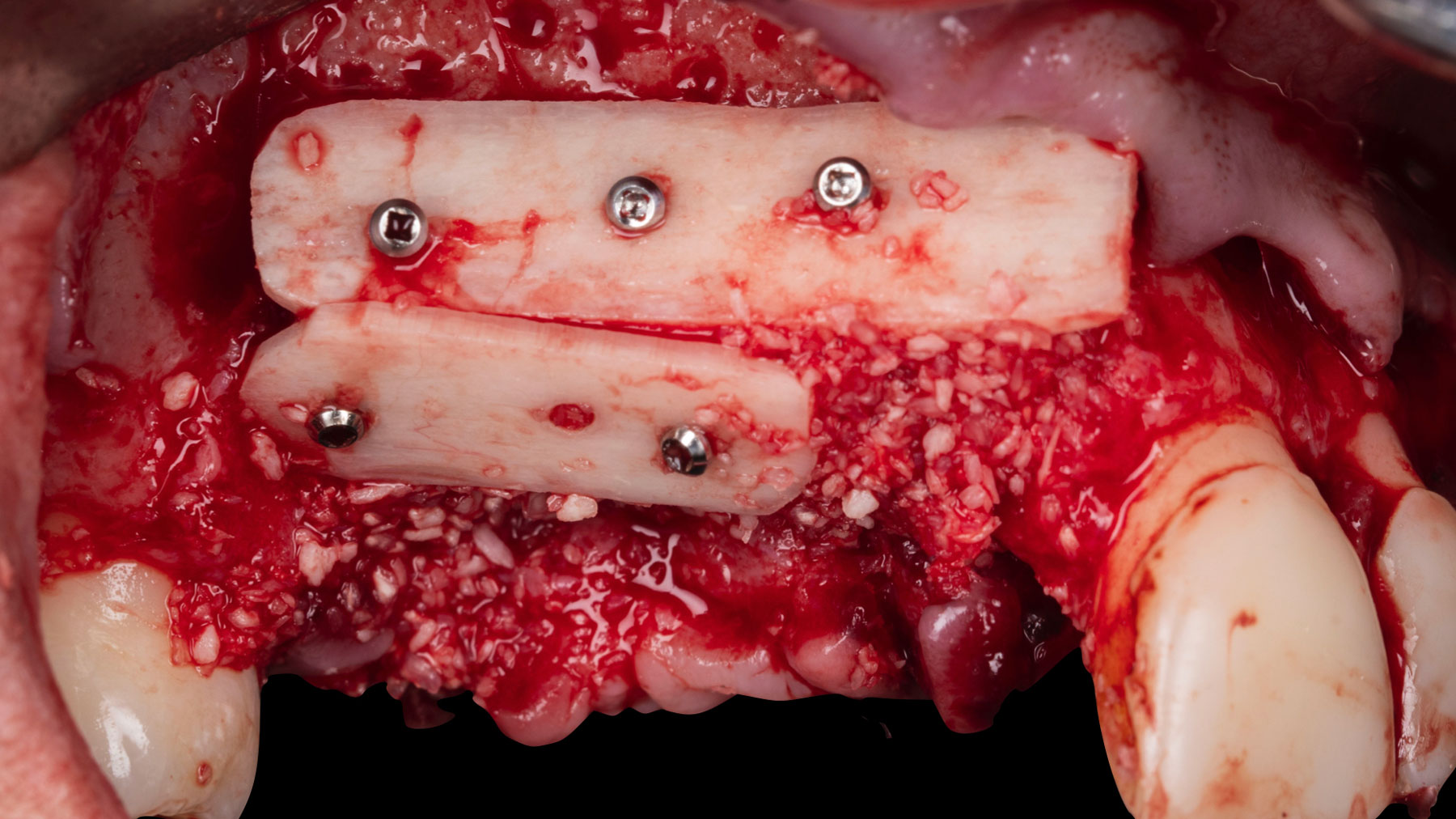
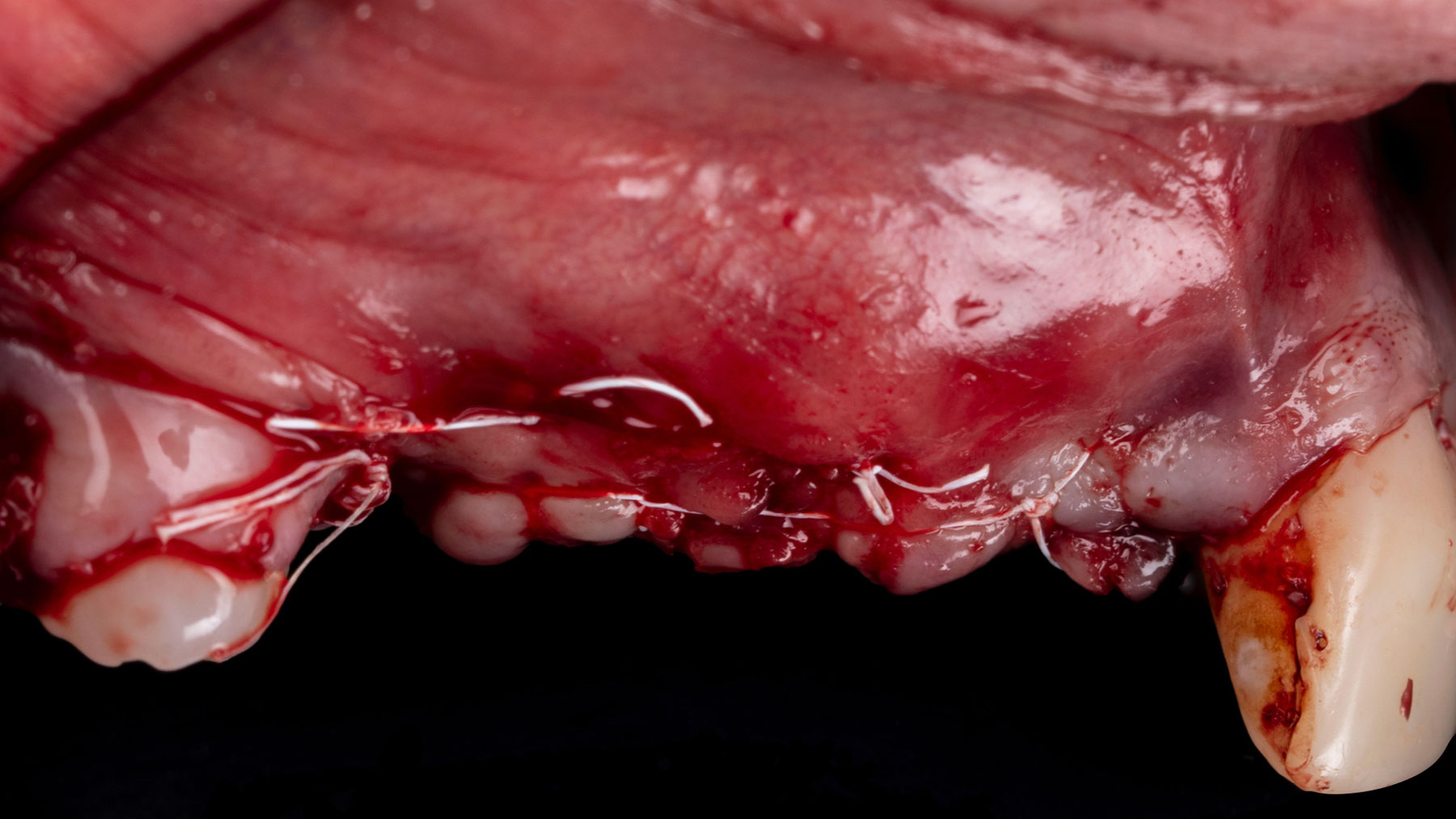
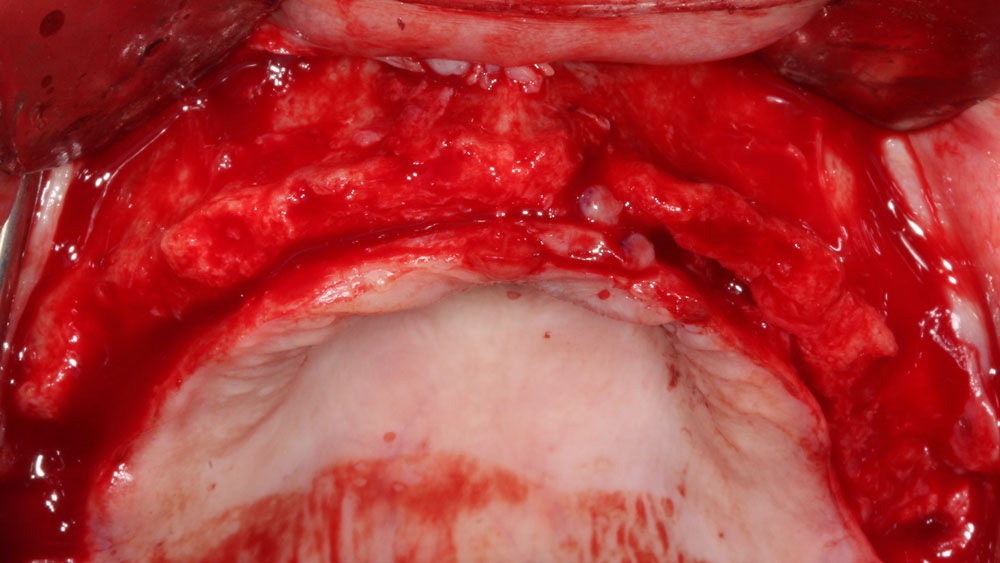
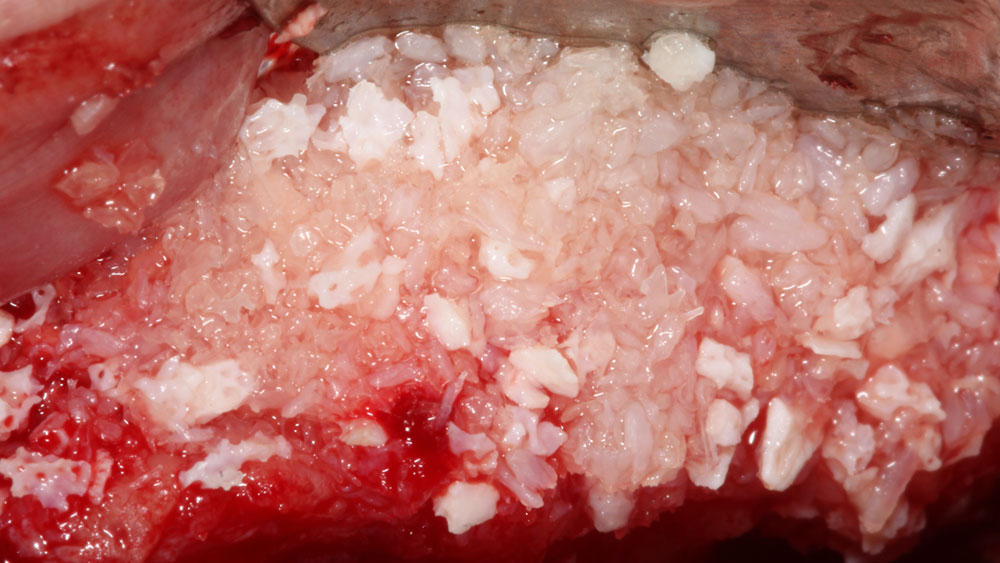
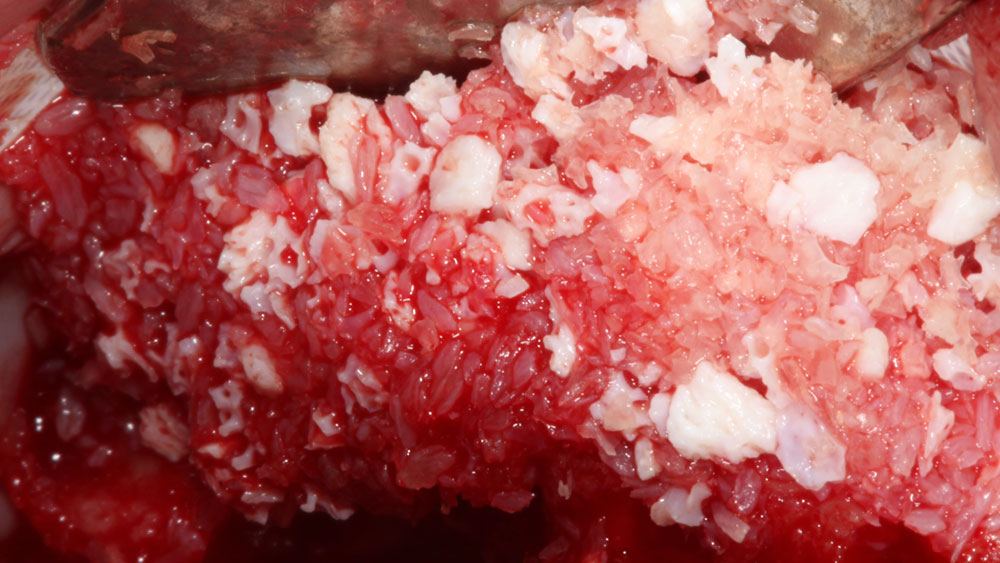
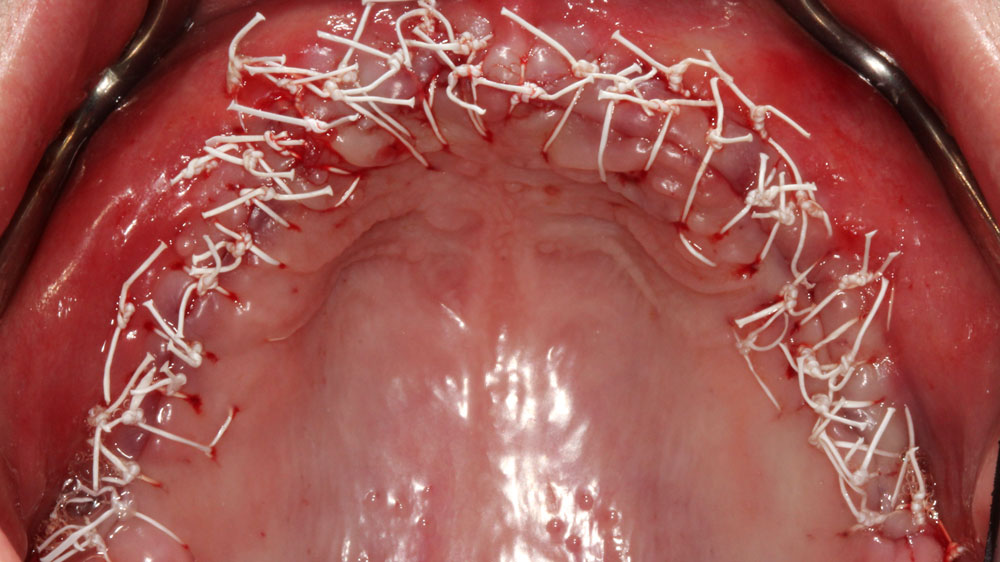
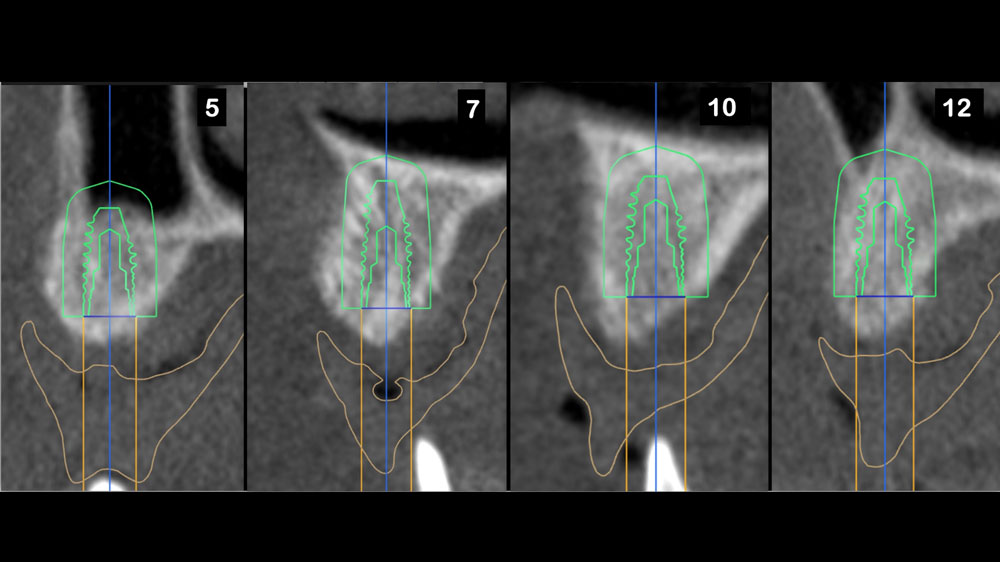
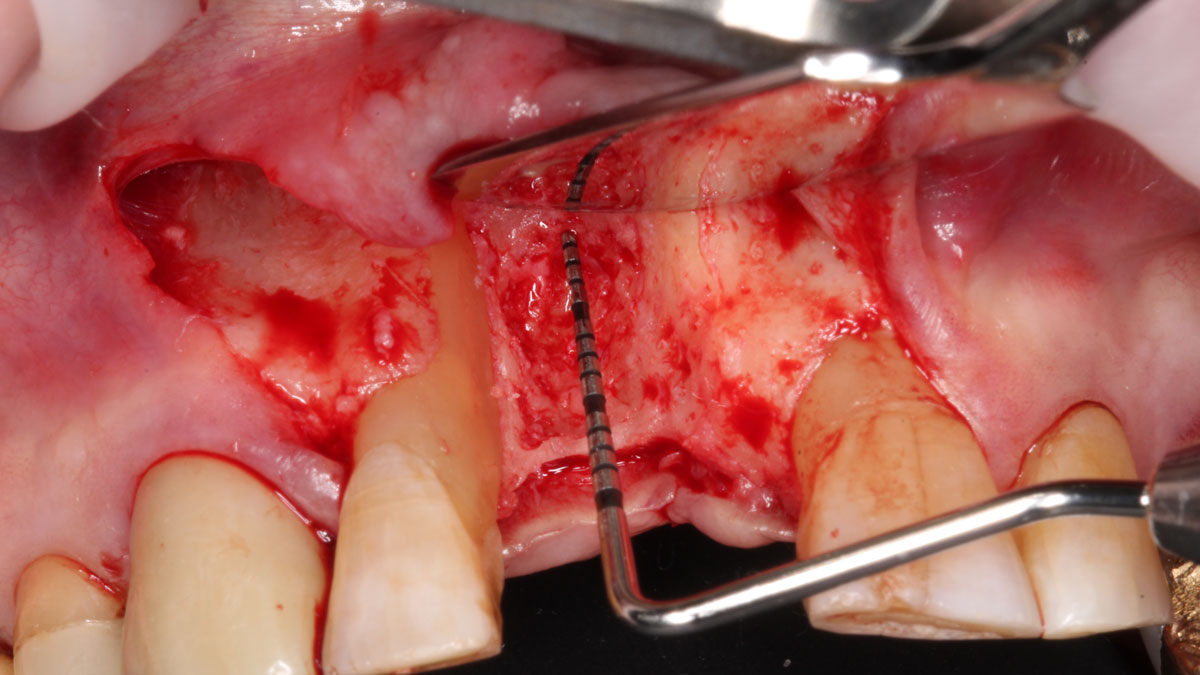
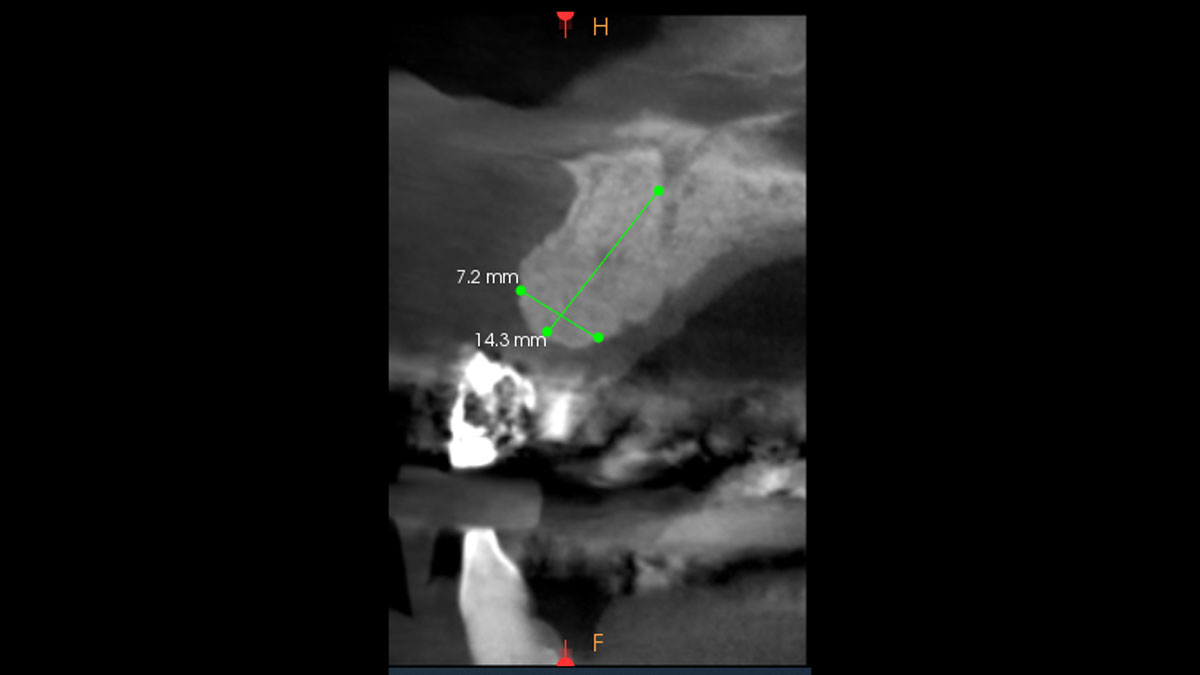
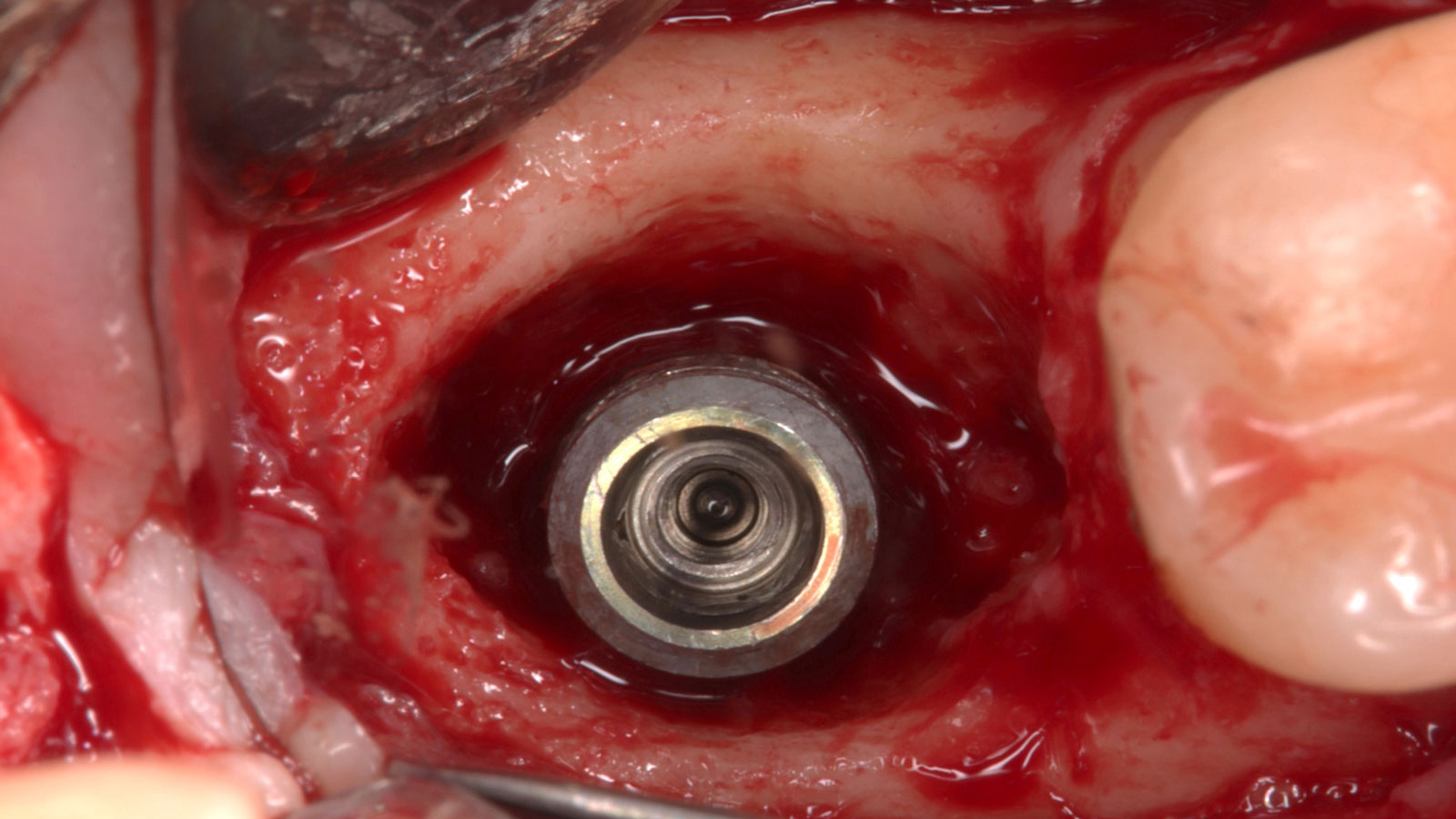
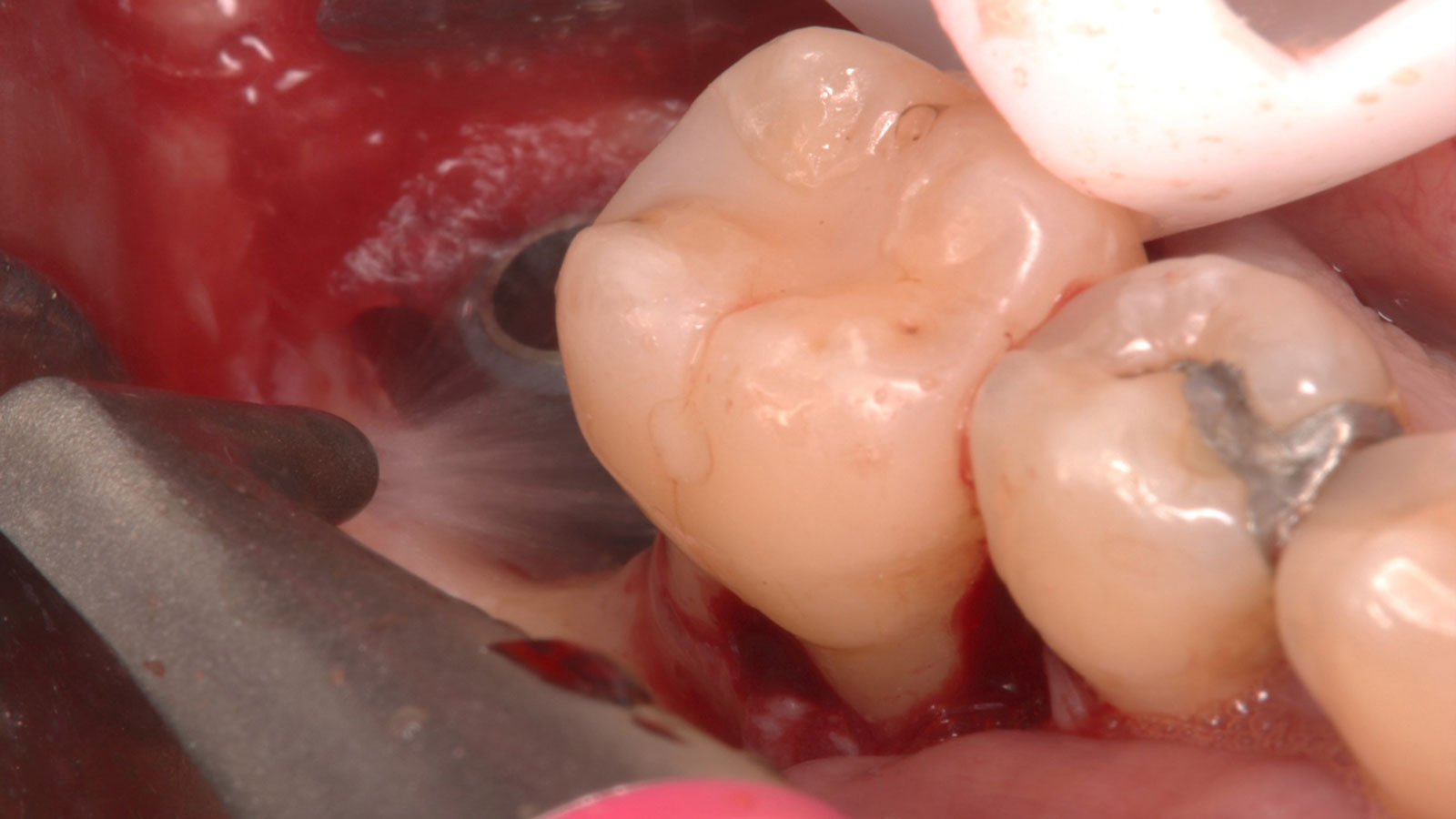
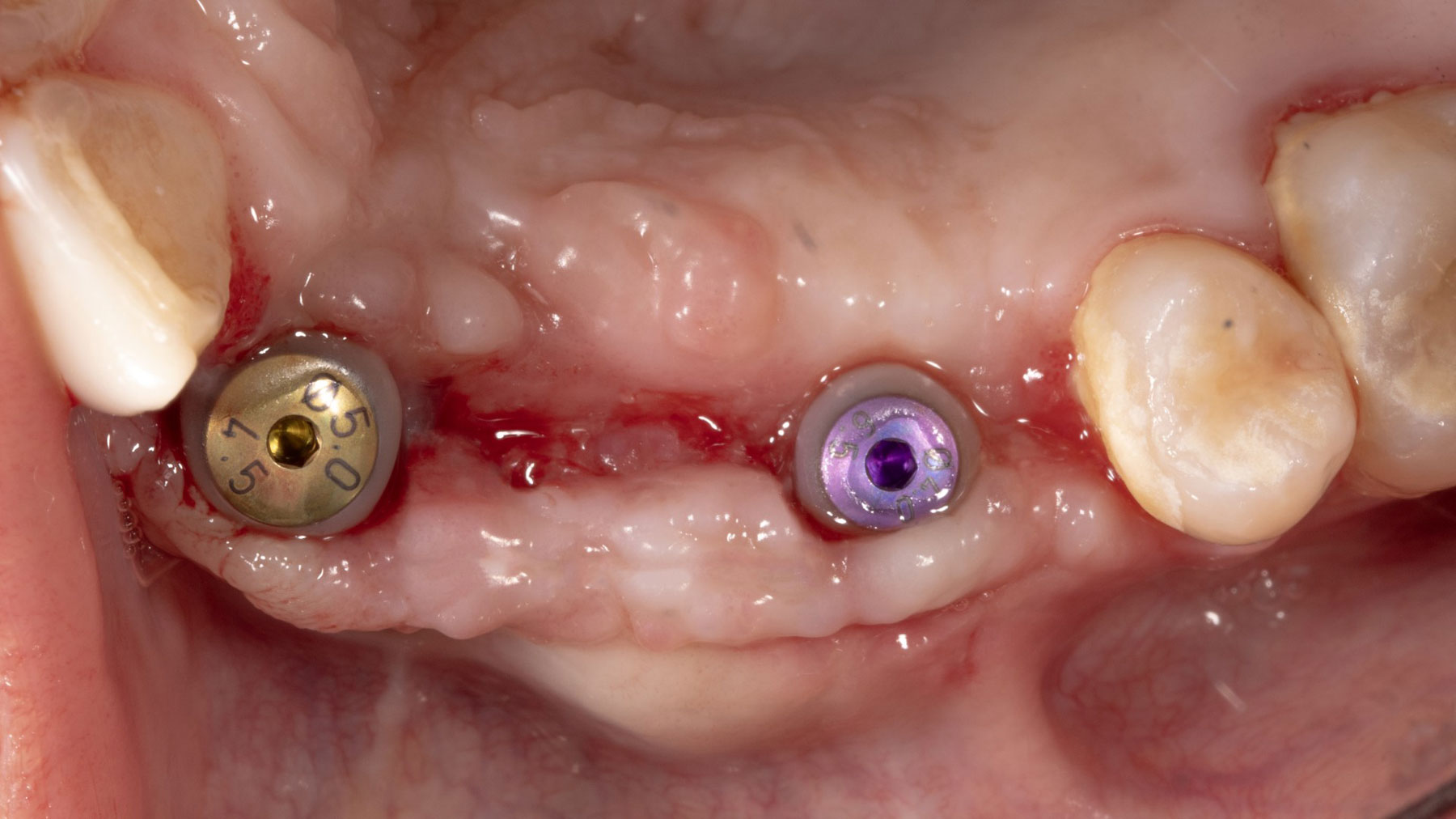
Vertical ridge augmentation was performed using allogenic cortical plates fixed with microscrews to create a stable, space-maintaining box, which was densely packed with vallos® mineralized cortico-cancellous granules hydrated with the rhPDGF-BB component of GEM 21S®. Five months later, the implants were placed, and the peri-implant soft tissue phenotype was enhanced using Geistlich Mucograft® and Geistlich Fibro-Gide® to improve soft-tissue height and thickness.
“The patient had high esthetic demands and specifically wanted to avoid autogenous grafting. Remarkably, this case was completed with 0% autogenous tissue, demonstrating that outcomes traditionally thought to require autogenous grafts can be achieved otherwise.”
— Dr. Muhammad Saleh
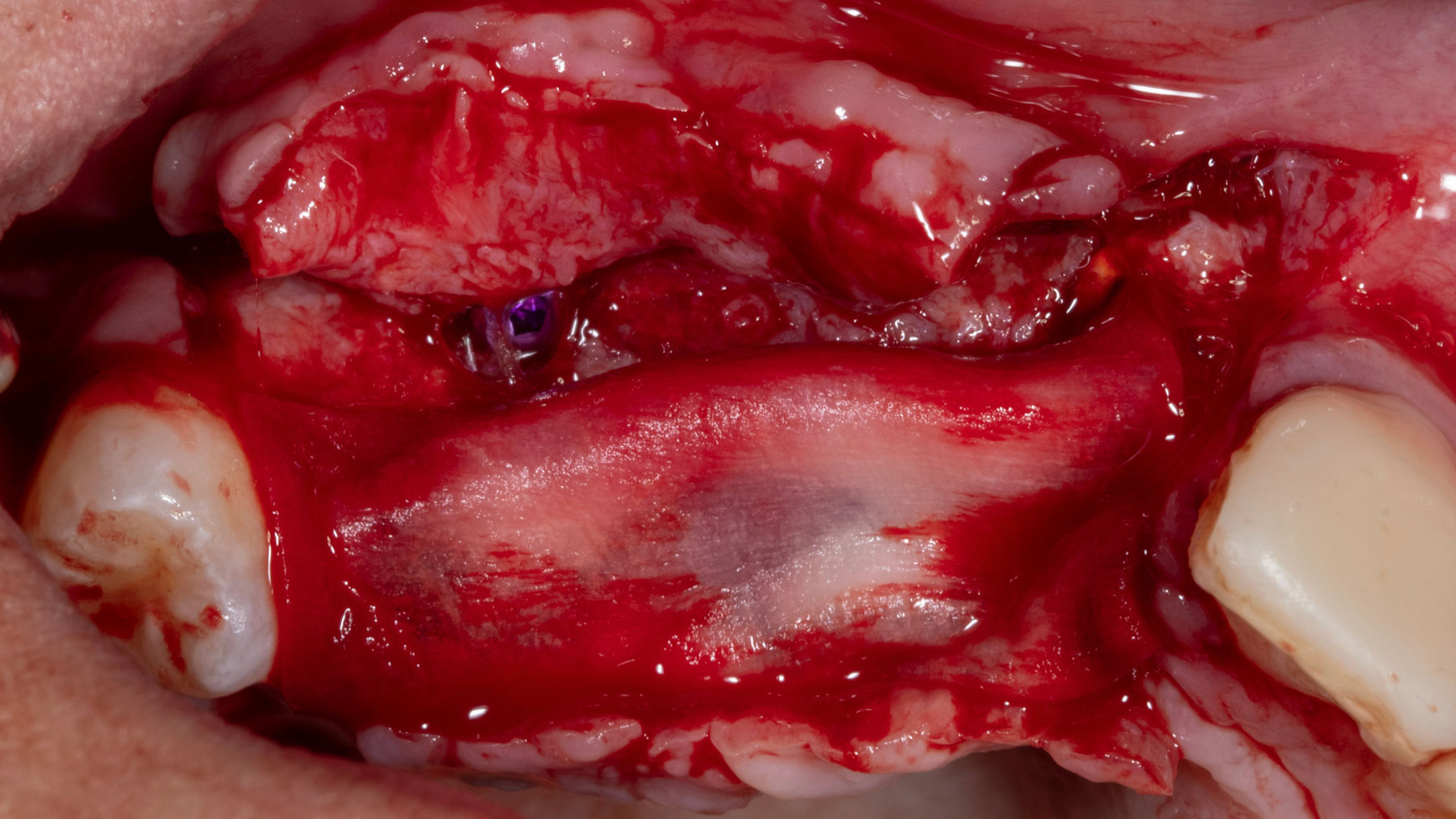
THE OUTCOME
Vertical ridge augmentation of the maxillary anterior was achieved using allogenic cortical bone plates fixed with microscrews to create a box configuration, filled with vallos® mineralized cortico-cancellous granules mixed with rhPDGF-BB component of GEM21S®. At implant placement, soft tissue phenotype was enhanced using Geistlich Mucograft® and Geistlich Fibro-Gide®.

Muhammad Saleh, BDS, MSD, PhD
Dr. Muhammad H. Saleh, Diplomate of the American Board of Periodontology, is a full-time faculty member at the University of Michigan and an internationally recognized lecturer in periodontology and implant dentistry. He has published over 140 peer-reviewed articles and reviews for leading Q1 journals. His numerous honors include the 2021 AAPF Schoor Award, 2022 Ramfjord Symposium Award, 2022 AAP Institute Award, 2024 AAPF LEAD and Nevins Awards, and the 2025 Roy H. Roberts Award.

BIOBRIEF
Vertical and Horizontal Maxillary Ridge Reconstruction with Advanced Grafting

THE SITUATION
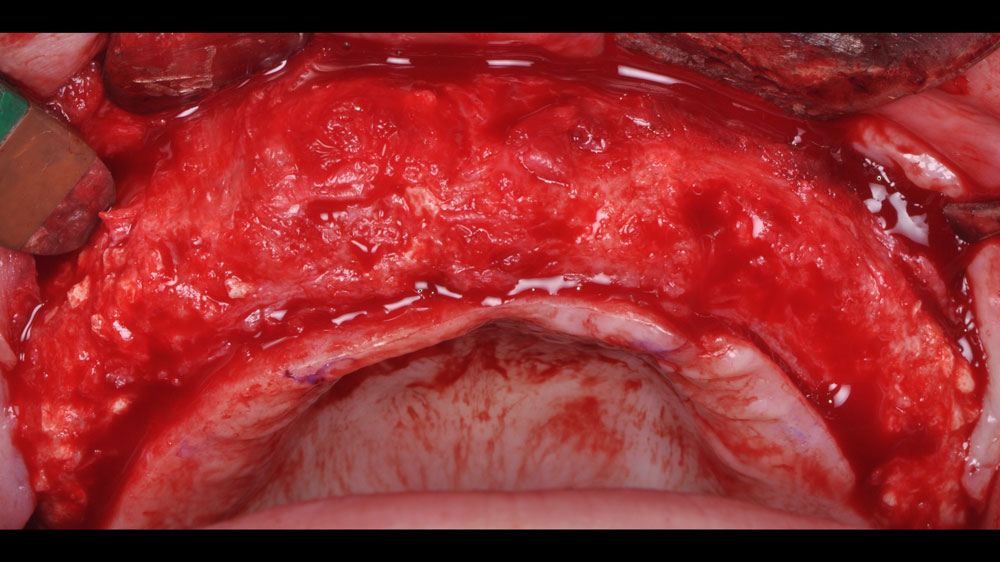
A 26-year-old edentulous female presented for implant-supported maxillary rehabilitation. She was systemically healthy and classified as ASA I. Clinical and radiographic evaluation revealed a Seibert Class III maxillary ridge defect with combined horizontal and vertical deficiencies. The patient exhibited a medium smile line and reported a highly active lifestyle with regular physical activity, indicating the need for a stable, durable, and esthetically driven implant rehabilitation capable of meeting long-term functional demands.
THE RISK PROFILE
| Low Risk | Medium Risk | High Risk | |
|---|---|---|---|
| Patient’s health | Intact immune system Non-smoker | Light smoker | Impaired immune system |
| Patient’s esthetic requirements | Low | Medium | High |
| Height of the smile line | Low | Medium | High |
| Gingival biotype | Thick – “low scalloped” | Medium – “medium scalloped” | Thin – “high scalloped” |
| Infection at implant sight | None | Chronic | Acute |
| Bone height at adjacent tooth | ≤ 5 mm from contact point | 5.5 – 6.5 mm from contact point | ≥ 7 mm from contact point |
| Soft-tissue anatomy | Intact | Compromised | |
| Bone anatomy of the alveolar ridge | No defect | Horizontal defect | Vertical defect |
THE APPROACH
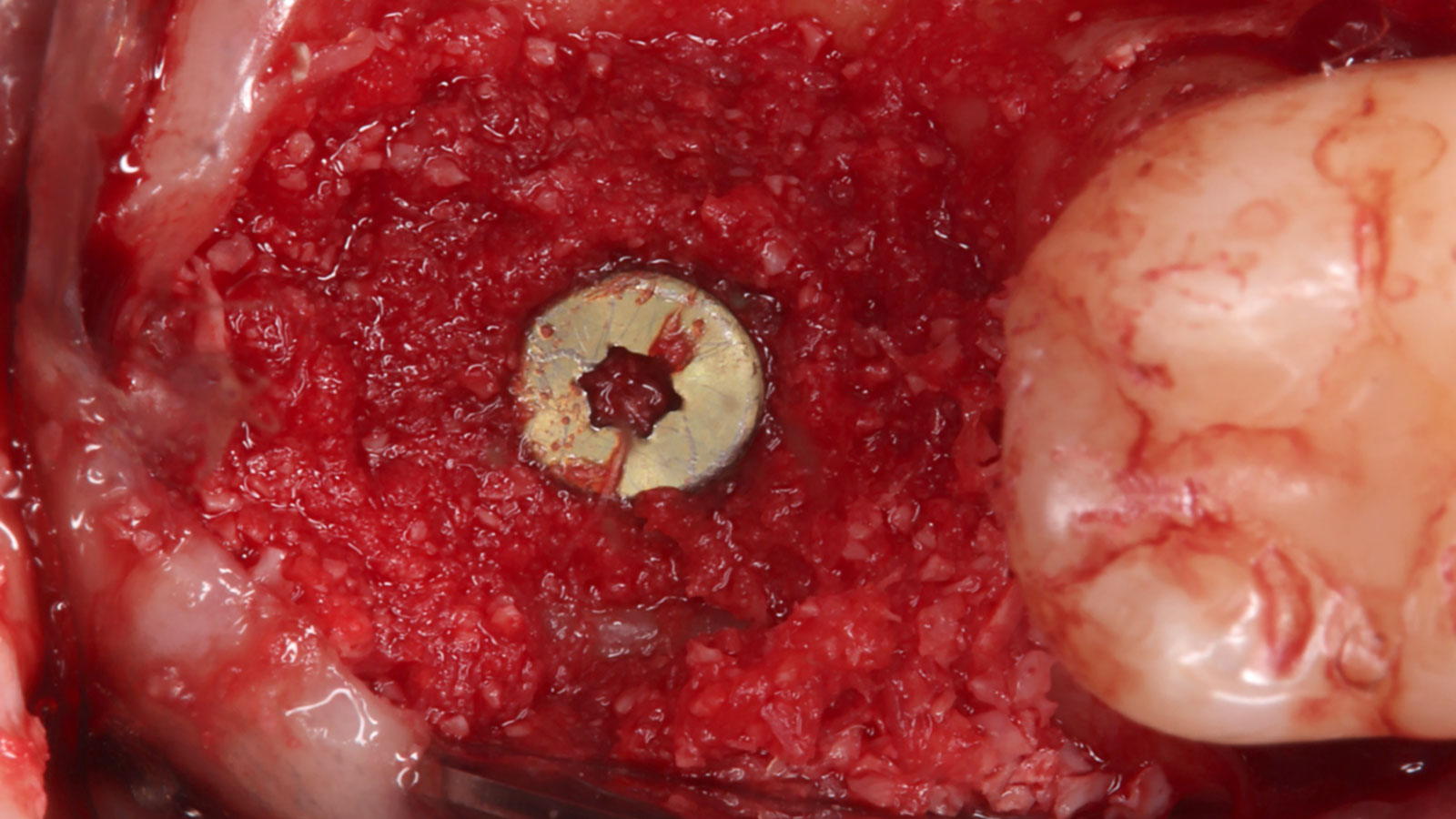
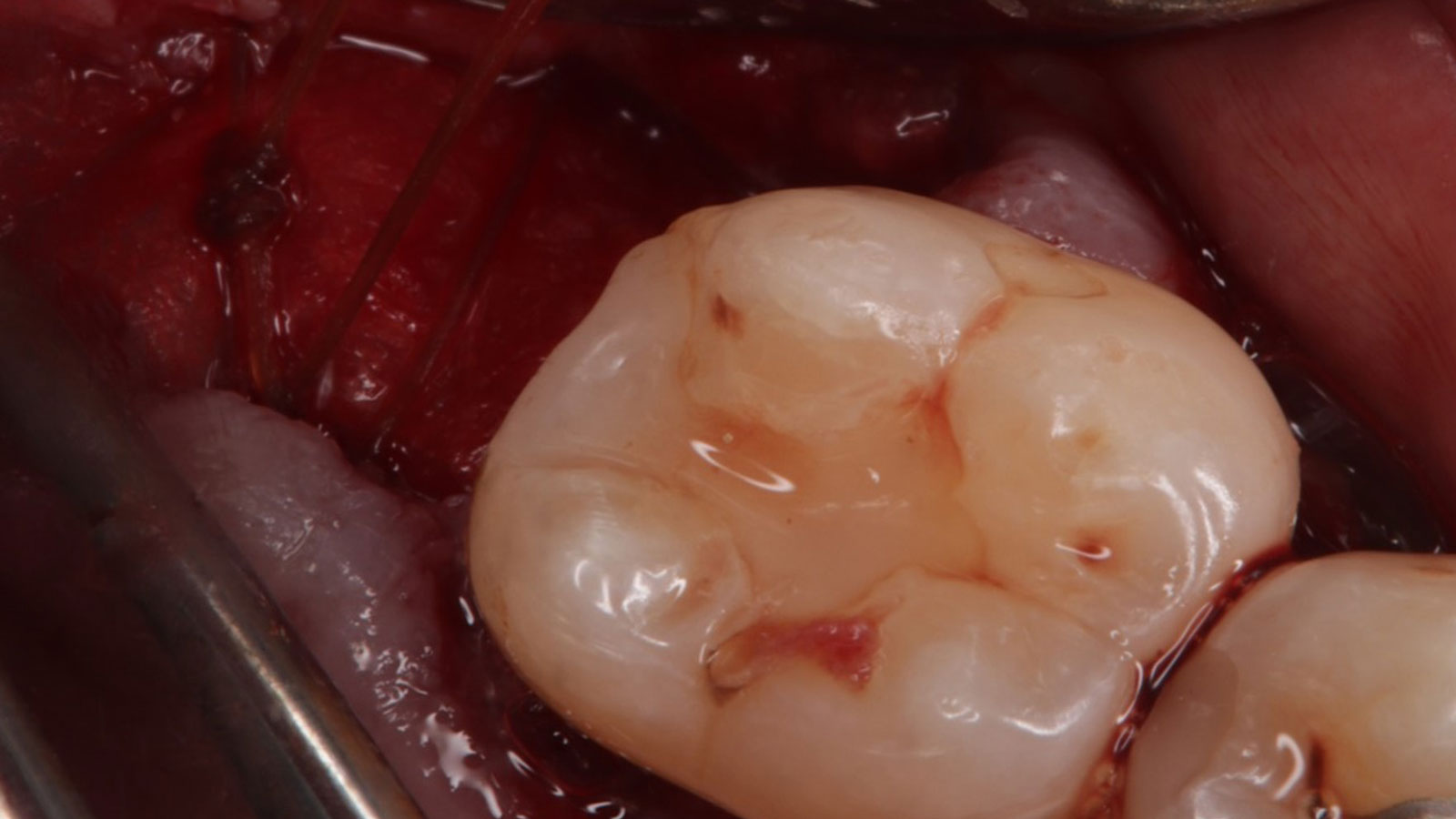
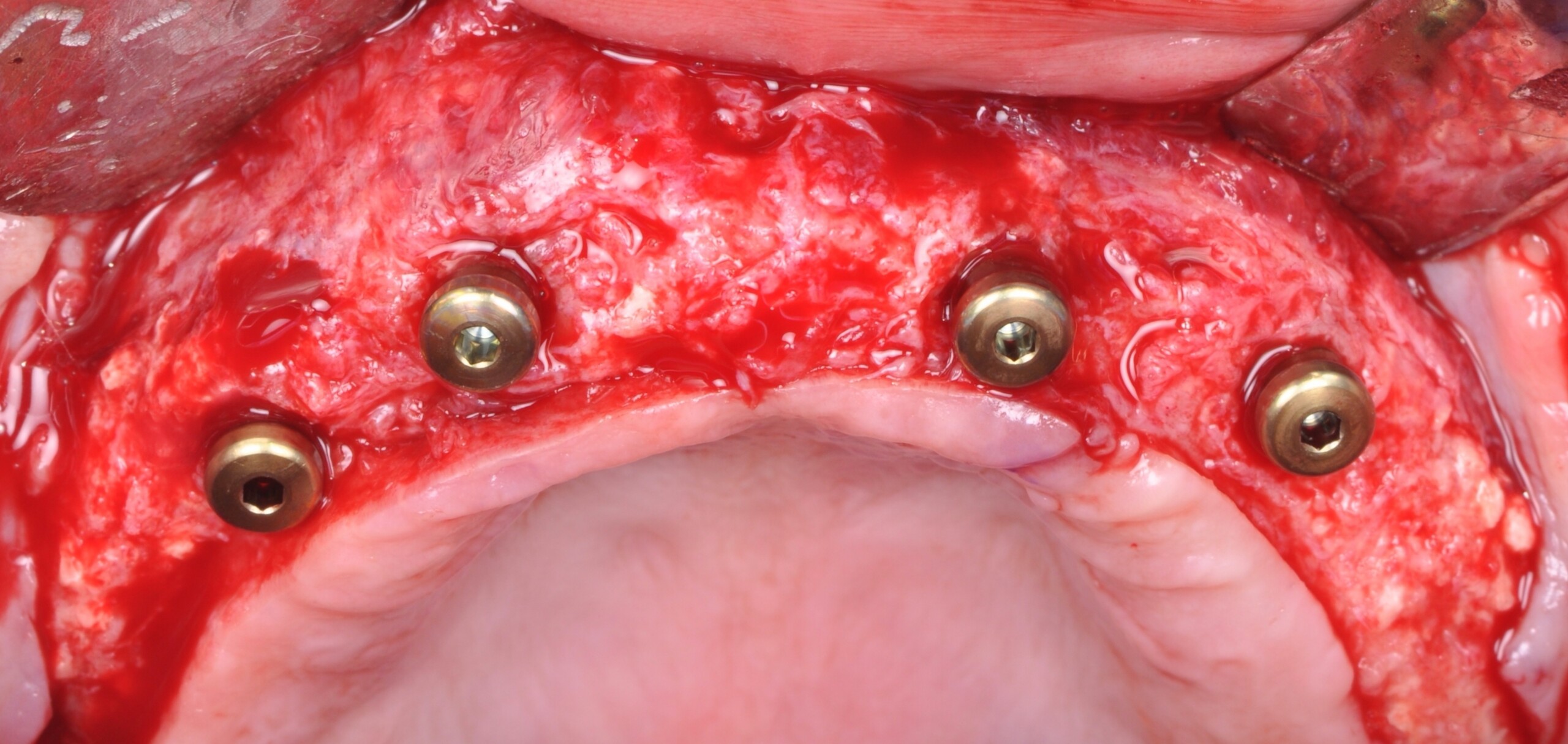
Horizontal and vertical ridge augmentation was performed using a full-thickness maxillary flap with two distal vertical releasing incisions. Extensive buccal periosteal release and cortical perforations were carried out. A composite graft of large-particle vallos® allograft (demineralized cortical granules) and large-particle Geistlich Bio-Oss® xenograft, combined with rhPDGF-BB, a component of GEM 21S®, was placed and stabilized under a high-density PTFE membrane, which was secured with four horizontal mattress and multiple simple interrupted 4-0 PTFE sutures.
“The patient’s young age and
— Nikolaos Soldatos, DDS, PhD, MSD
excellent systemic health are favorable prognostic factors, but achieving optimal outcomes requires meticulous surgical execution, careful soft tissue management, and strict adherence to post-operative instructions to minimize complications and ensure long-term success.”
THE OUTCOME
Horizontal (5–7 mm) and vertical (3–4 mm) ridge augmentation were successfully obtained, with stablewound management achieved through precise suturing techniques. The osteoinductive properties of the large vallos®, demineralized granules combined with rhPDGF-BB, a component of GEM21S® promoted high-quality bone regeneration, whereas the large Geistlich Bio‑Oss®, xenograft particles contributed to volume preservation by moderating resorption. As a result, four 4.3 mm implants were placed with high primary stability, each exceeding 35 N·cm of insertion torque, providing a strong foundation for a predictable esthetic and functional restoration.

Nikolaos Soldatos, DDS, PhD, MSD
Dr. Soldatos is a tenure-track Associate Professor and Clinical Director of the Postgraduate Advanced Program in Periodontics at Oregon Health & Science University. He is a Board-Certified Periodontist and Implant Surgeon, he holds a DDS, PhD, and MSD, and has completed advanced periodontal and implant training in both the U.S. and Europe. Dr. Soldatos’ research focuses on translational implant biology and bone regeneration. He is also a Fellow of the Academy of Osseointegration.

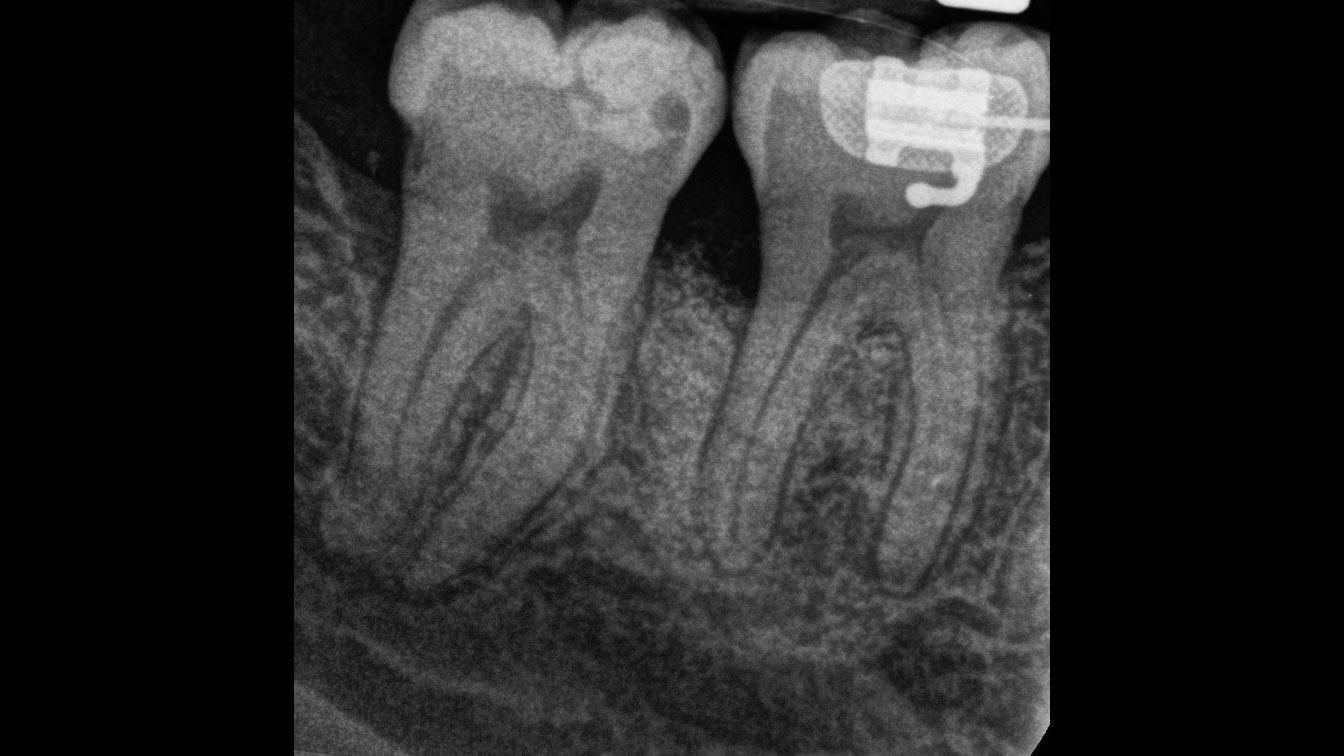
CLINICAL CASE
THE OUTCOME
At 6 months, healing was complete with a bone fill of 10x10x6.5 mm. Tooth #7, affected by the combined defect, was fully regenerated. The adjacent ridge defect was also restored, enabling the placement of a regular-sized implant without the need for additional grafting.
Disclaimer: These results are not guaranteed; individual outcomes may vary depending on patient circumstances. This information is for informational purposes only and may not reflect Geistlich’s official position, opinion, or recommendation. Treatment decisions are made at the physician’s discretion, based on the unique needs of each patient.
GEM 21S® has not been approved by FDA for use in ridge augmentation in the US and the safety and effectiveness of GEM 21S® for this use has not been established by FDA.

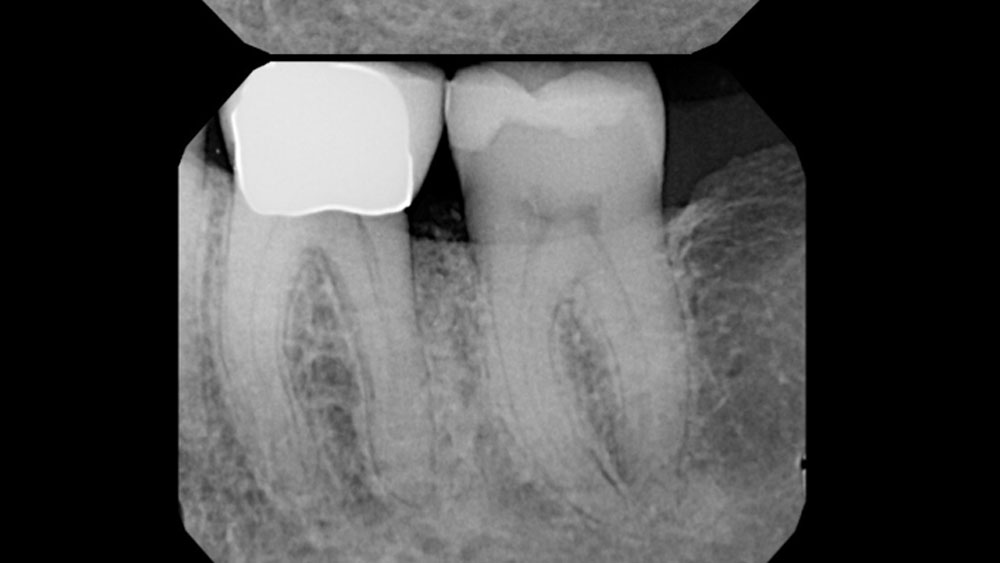
CLINICAL CASE
THE APPROACH
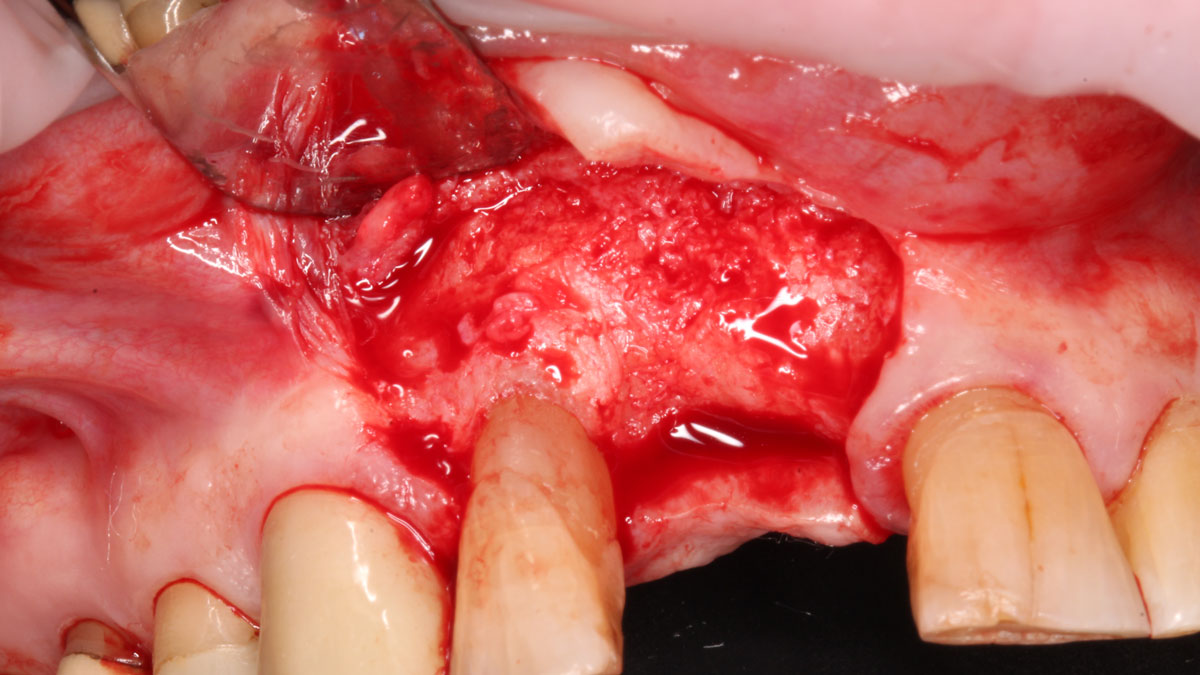
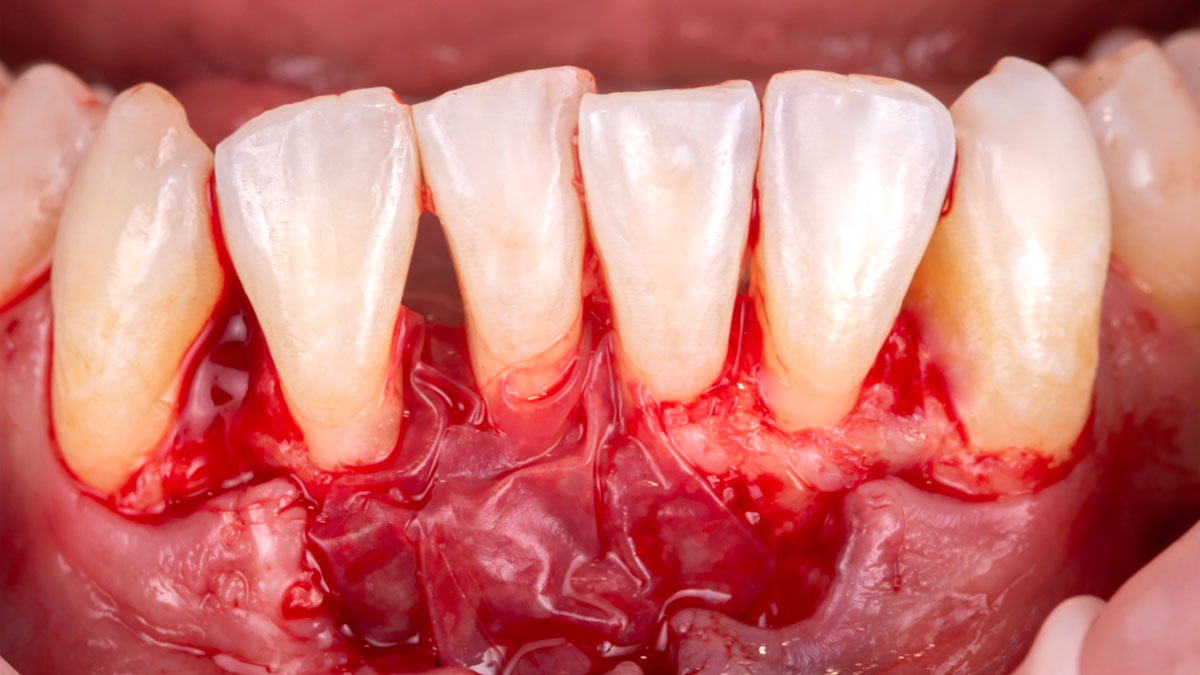
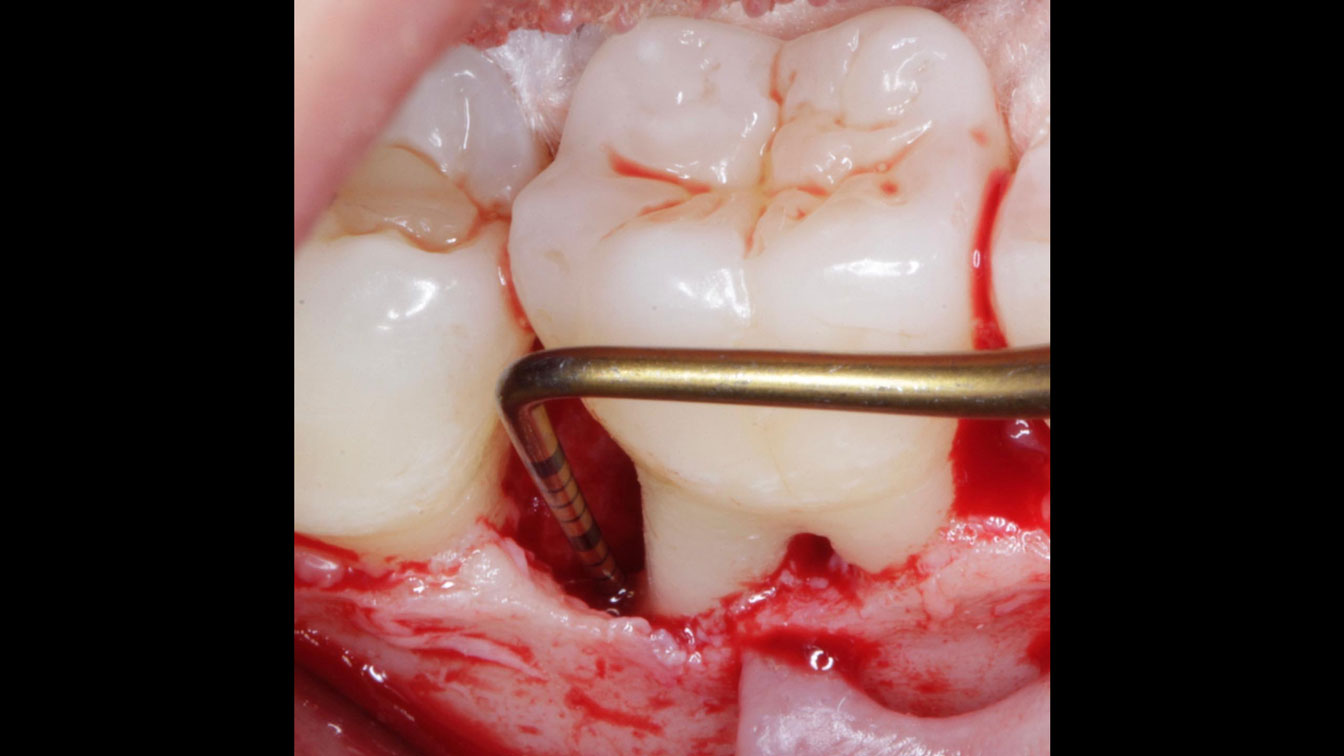
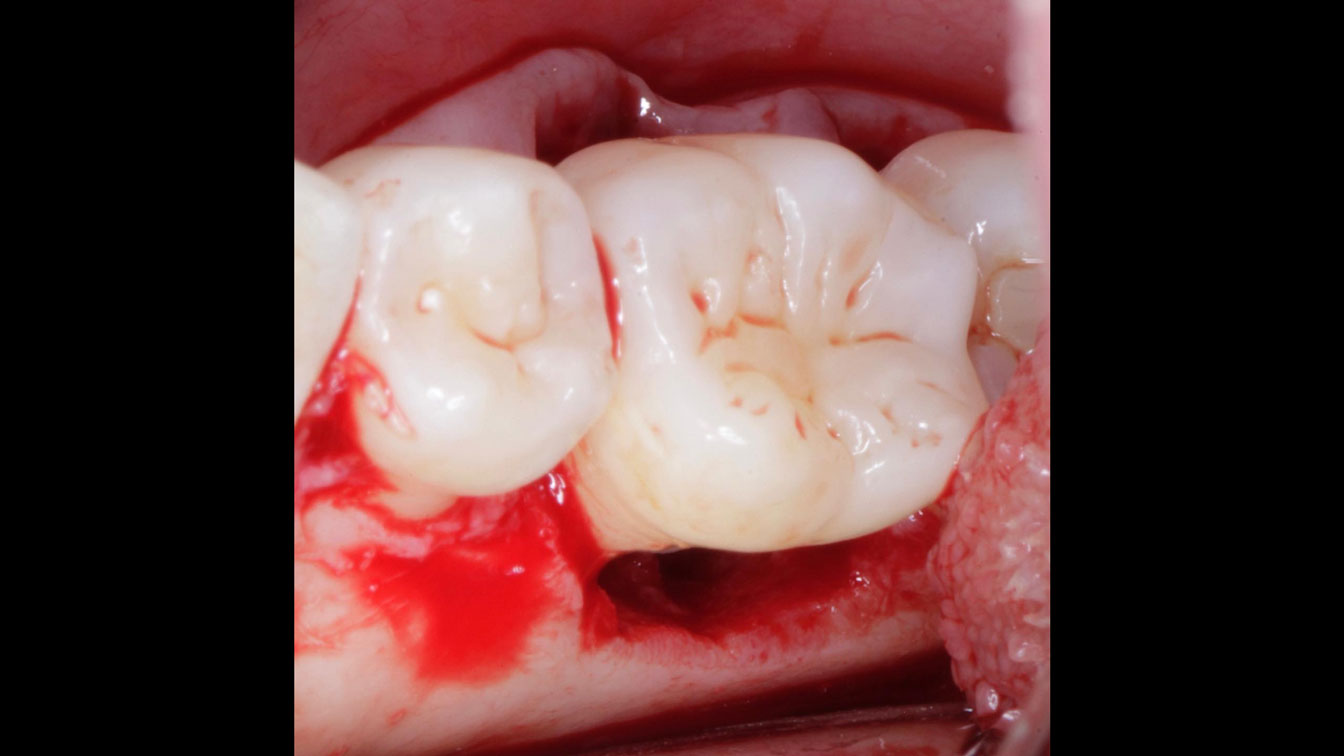
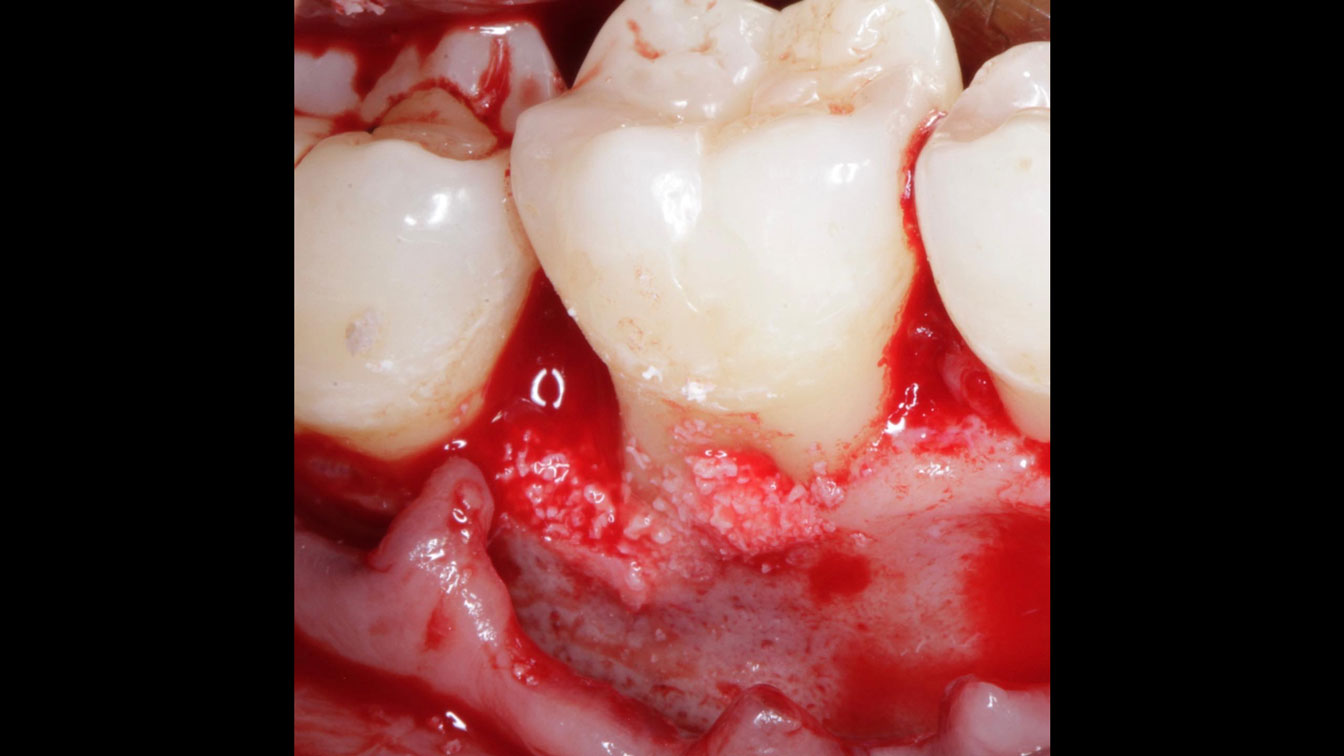
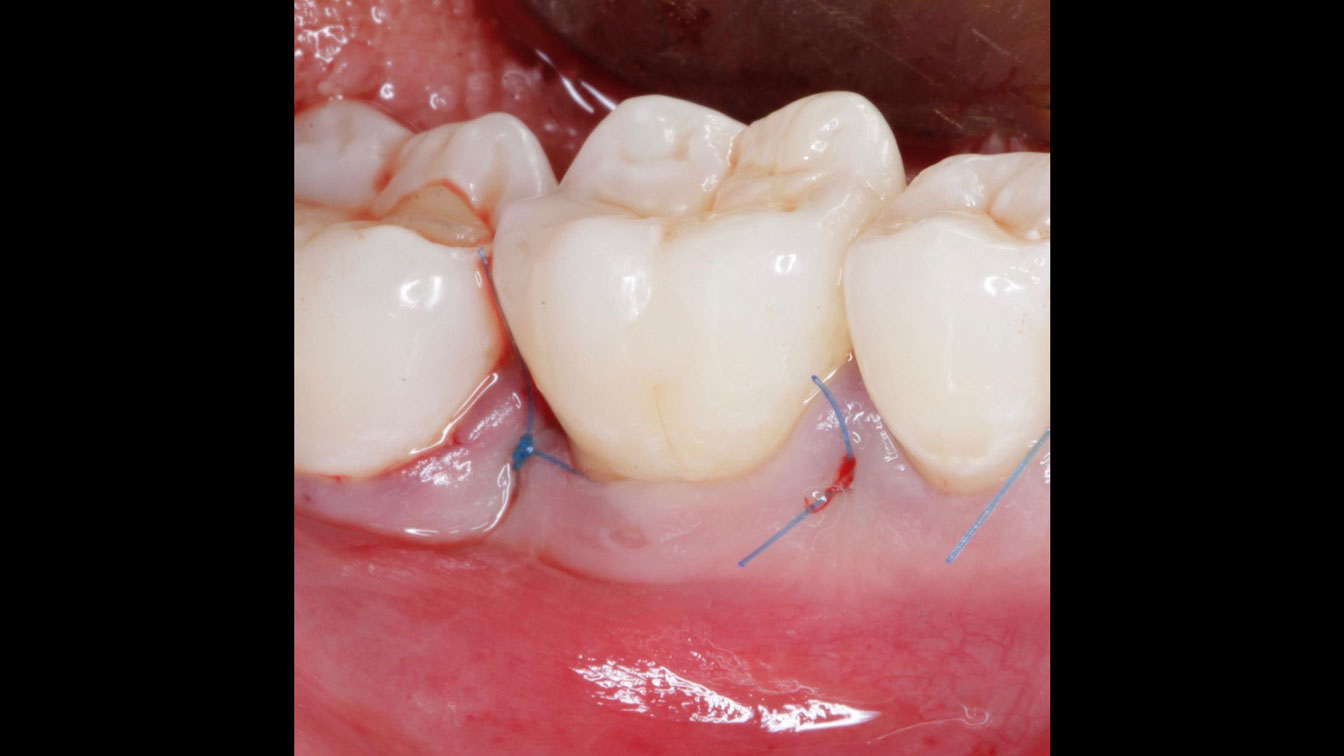
Considering the patient’s age, condition, and good compliance, a regenerative approach was chosen to preserve all teeth. Due to the extent and severity of the defect, a bone graft, biologics, and a membrane were utilized. The goal was to restore lost attachment and stabilize the teeth through splinted therapy.
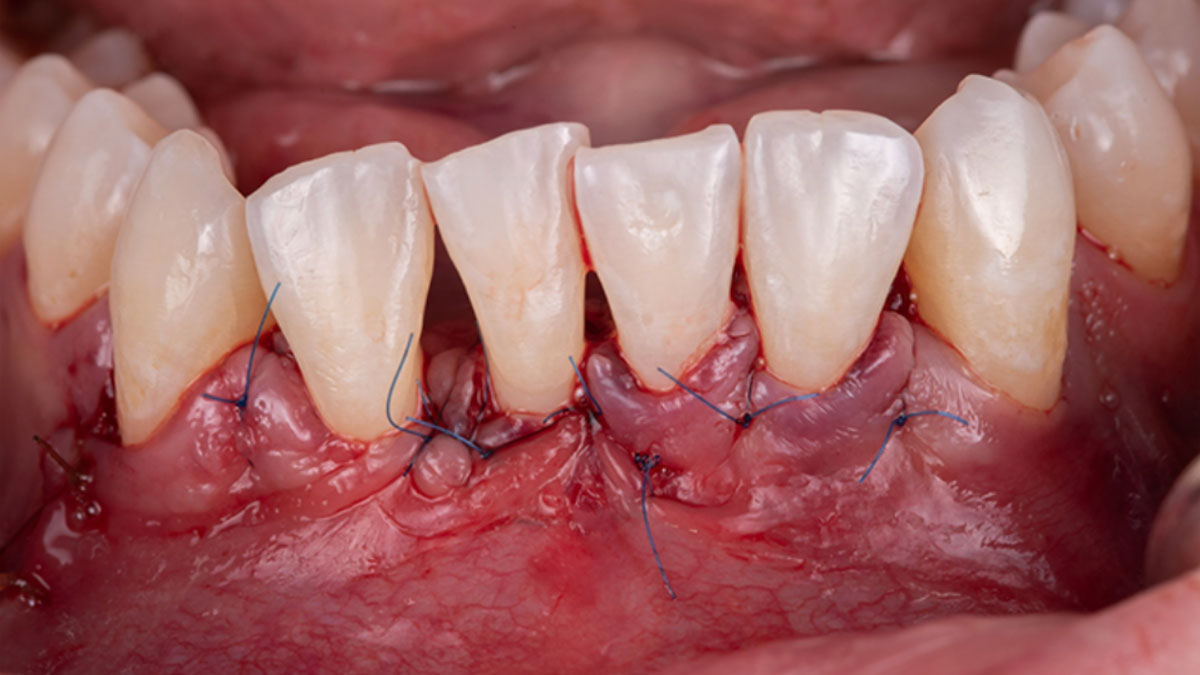
THE OUTCOME
At the 7-month follow-up, probing depths decreased from 7 mm (MB, DB) to 3 mm, with radiographic bone fill indicating a 90% success. Attachment gain of 3 mm was achieved, improving from 7 mm to 4 mm. The patient reported satisfactory outcomes, and splinting effectively maintained tooth stability with no mobility.
Disclaimer: These results are not guaranteed; individual outcomes may vary depending on patient circumstances. This information is for informational purposes only and may not reflect Geistlich’s official position, opinion, or recommendation. Treatment decisions are made at the physician’s discretion, based on the unique needs of each patient.

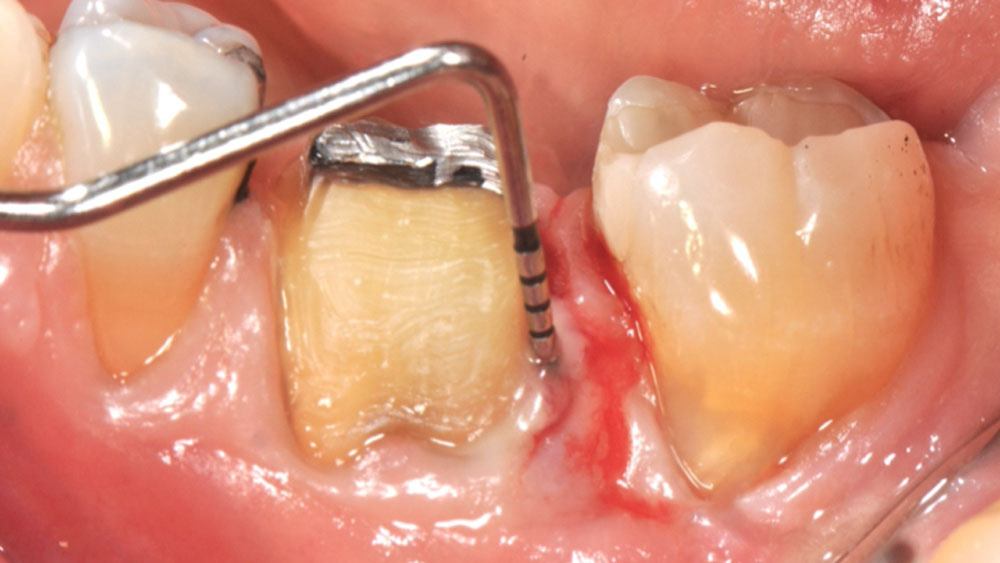
CLINICAL CASE
THE APPROACH
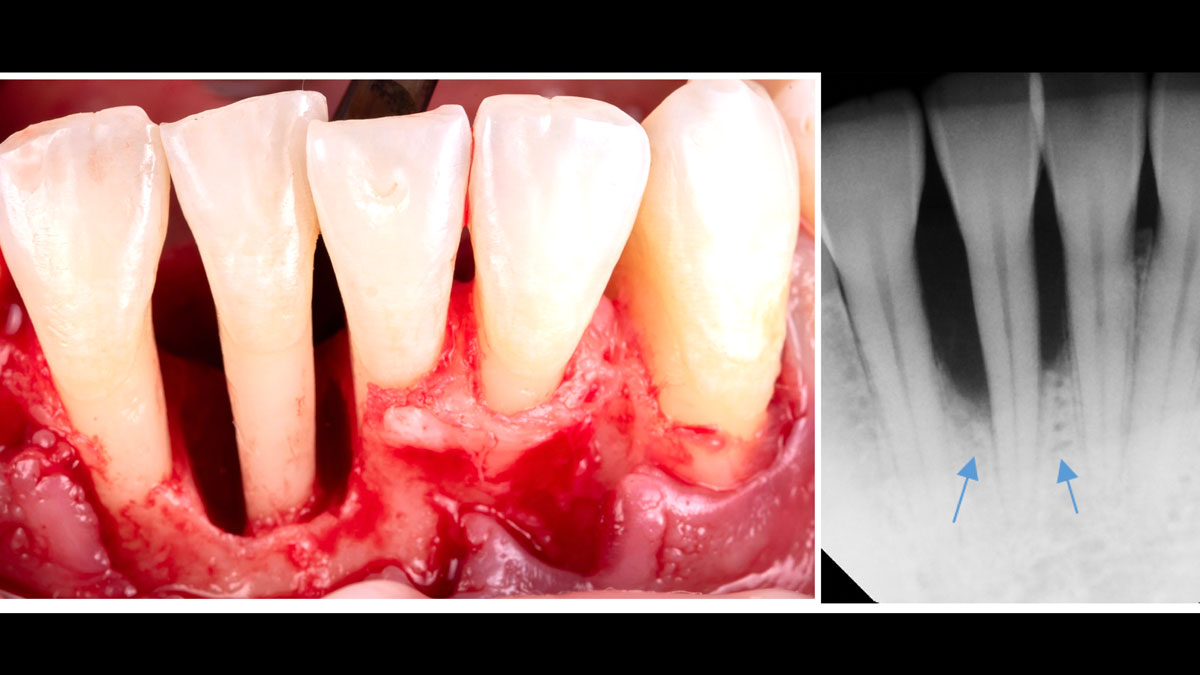
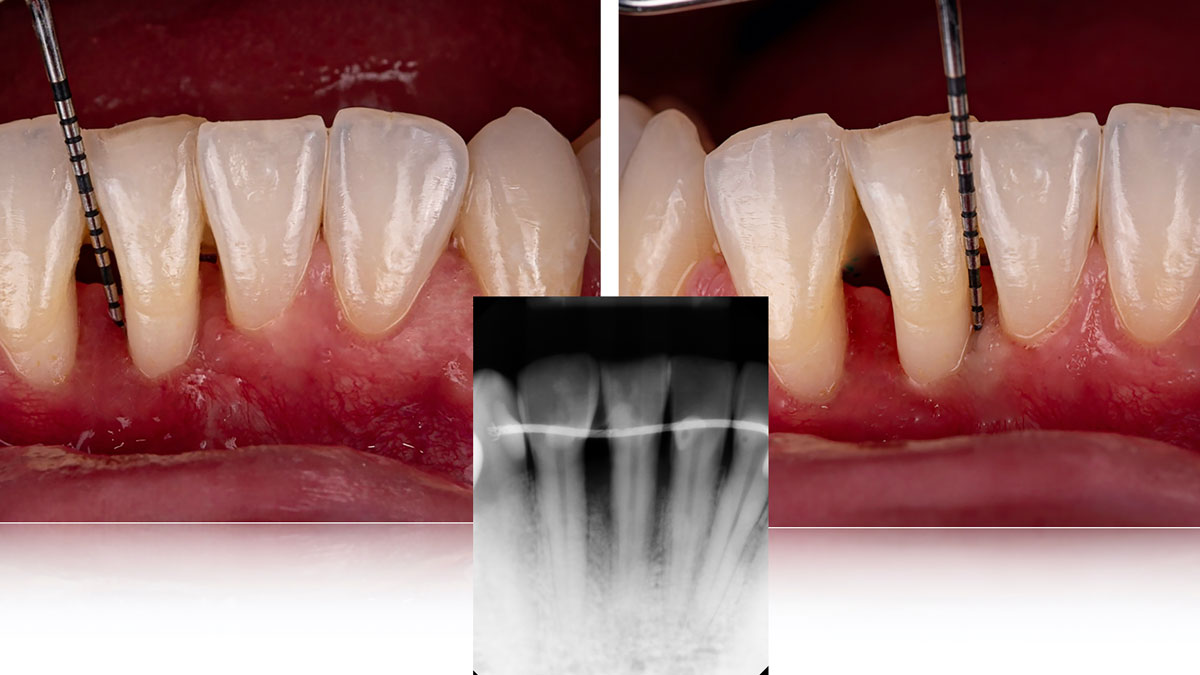
Following flap elevation and thorough defect debridement, a xenograft bone substitute mixed with PDGF (GEM 21S®) was used to fill the defect. The graft was left uncovered without a membrane.
THE OUTCOME
The 1-year follow-up radiograph demonstrated near-complete bone fill in a previously deep defect. Clinically, shallow probing depths and healthy tissue were also observed at the 1-year follow-up.
Disclaimer: These results are not guaranteed; individual outcomes may vary depending on patient circumstances. This information is for informational purposes only and may not reflect Geistlich’s official position, opinion, or recommendation. Treatment decisions are made at the physician’s discretion, based on the unique needs of each patient.

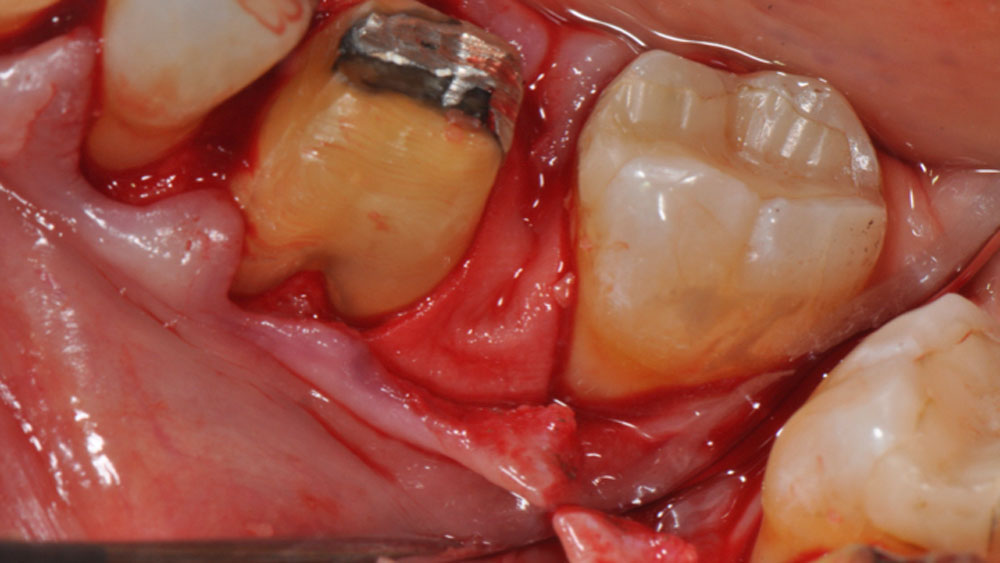
CLINICAL CASE
THE APPROACH
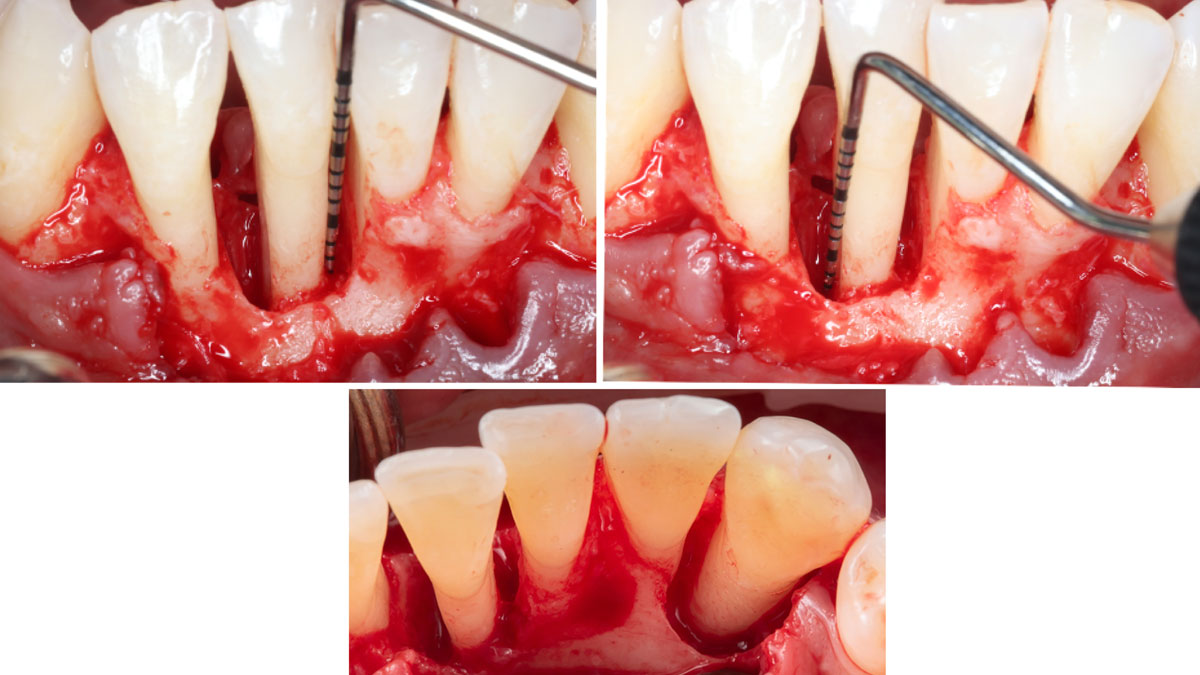
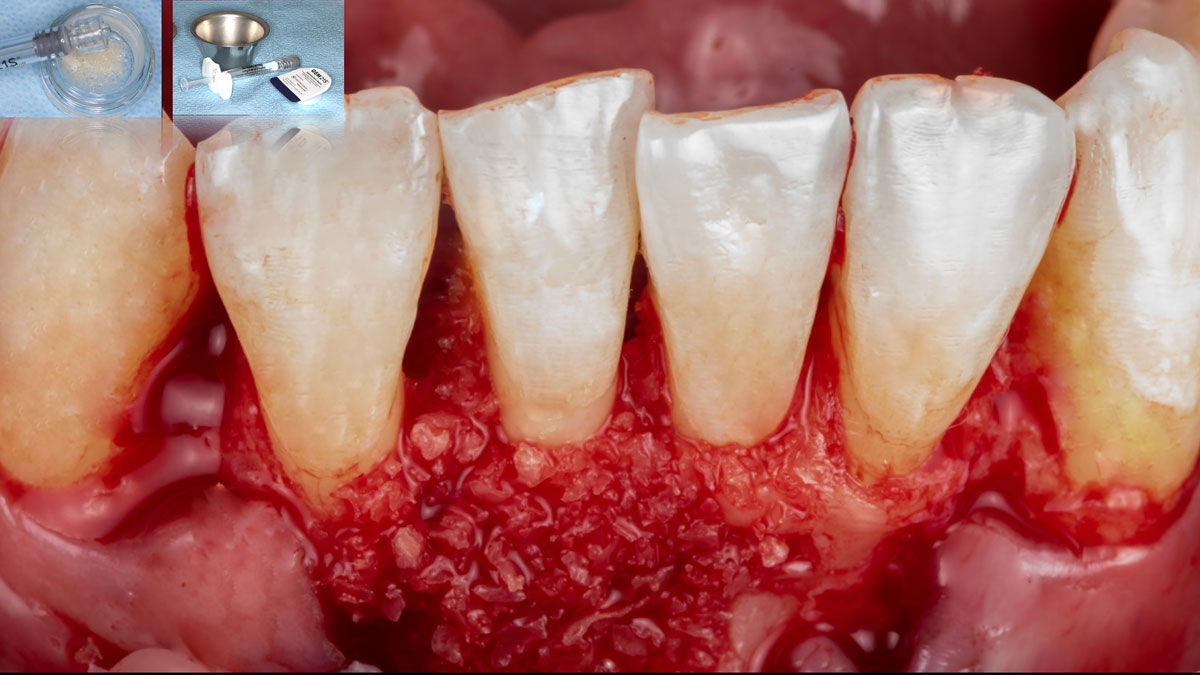
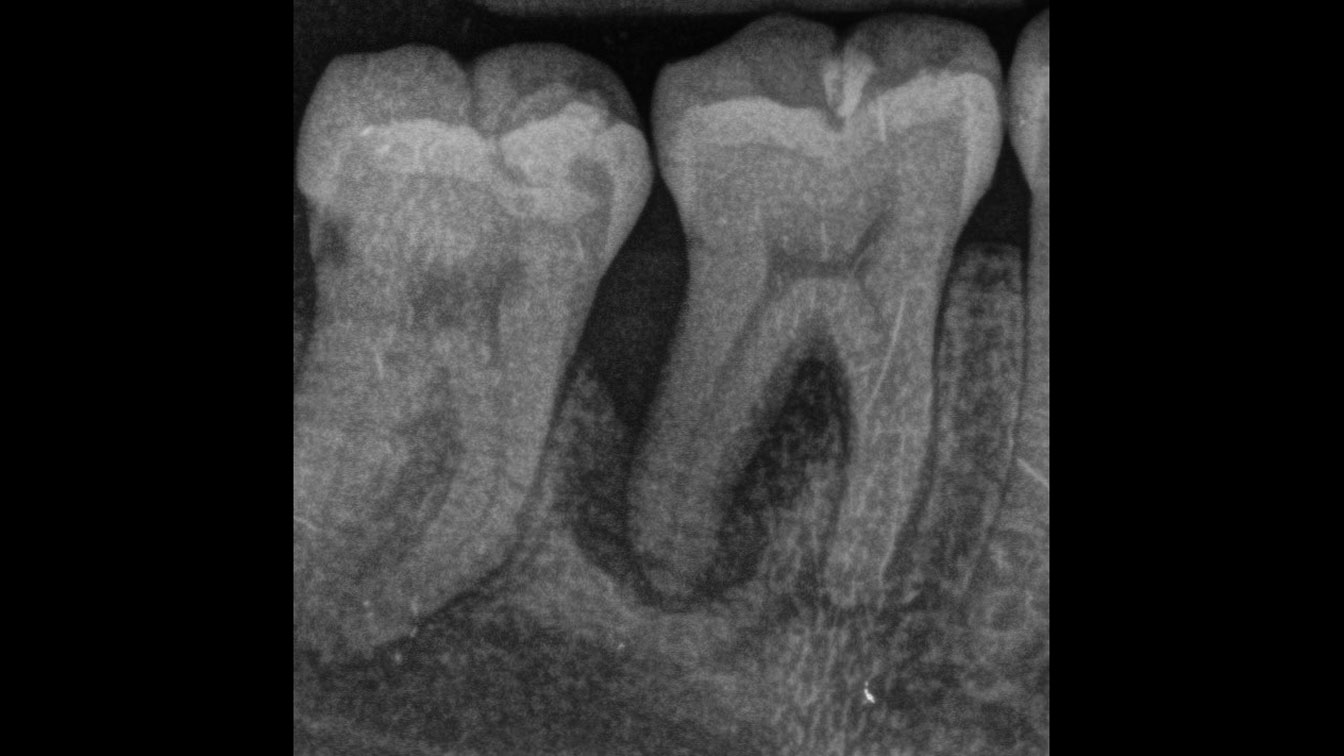
The crown was biologically shaped, and the root surface was detoxified using Ellman burs. Following flap elevation and thorough defect debridement, an allograft mixed with GEM 21S® was used to fill the defect. The graft was then covered with Geistlich Bio-Gide®.
THE OUTCOME
At the 10-month follow-up, radiographs revealed evidence of bone fill, and clinically, the interdental tissue showed signs of maturation. At the 9-year follow-up, clinical photos demonstrated long-term stability, with maintained bone levels, minimal interproximal recession, and lack of facial recession. Radiographic analysis further confirmed the sustained stability of the bone.
Disclaimer: These results are not guaranteed; individual outcomes may vary depending on patient circumstances. This information is for informational purposes only and may not reflect Geistlich’s official position, opinion, or recommendation. Treatment decisions are made at the physician’s discretion, based on the unique needs of each patient.

BIOBRIEF
Prosthetic-Surgical Approach to Regenerative Treatment for Peri-Implantitis
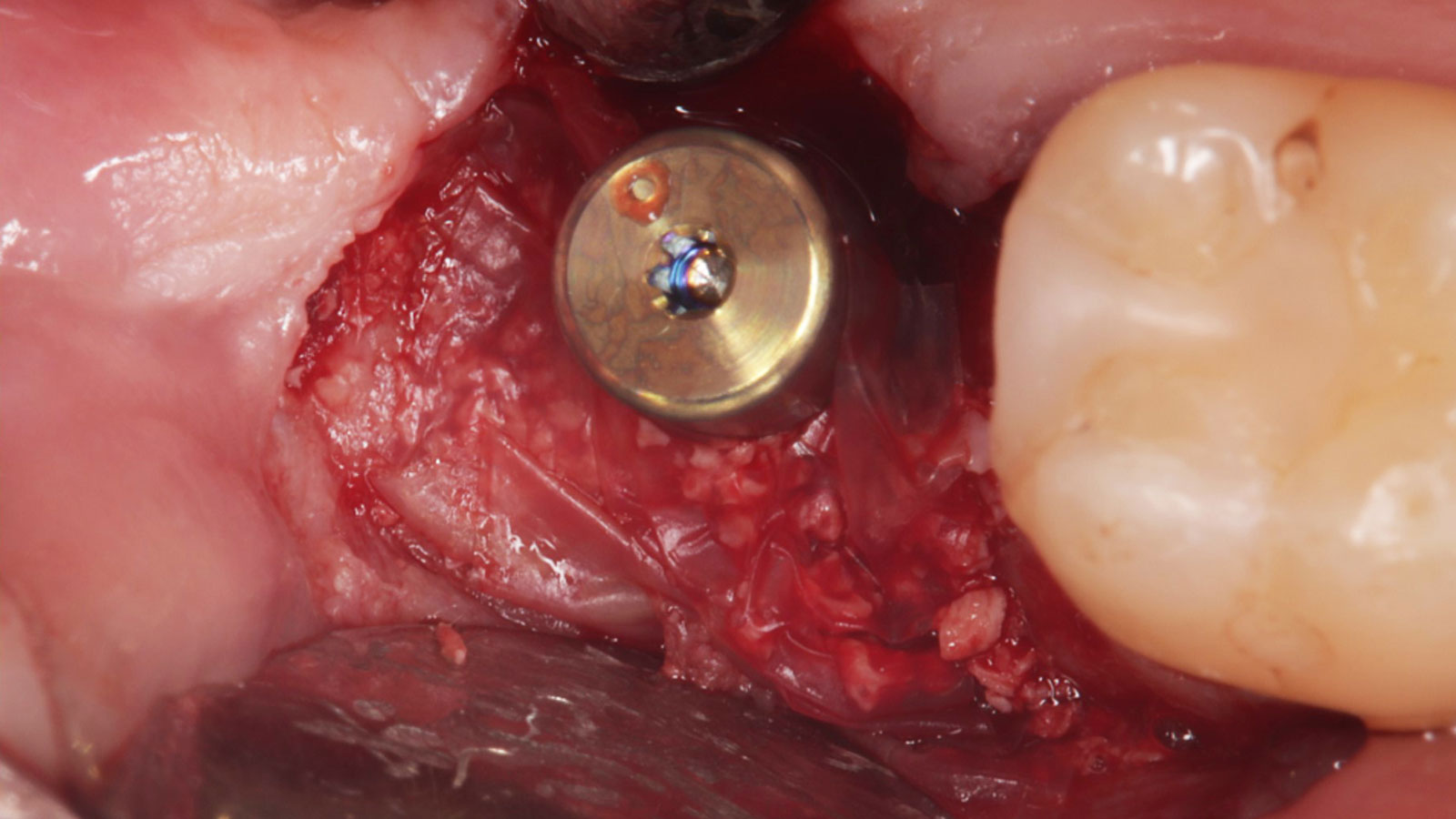


THE SITUATION
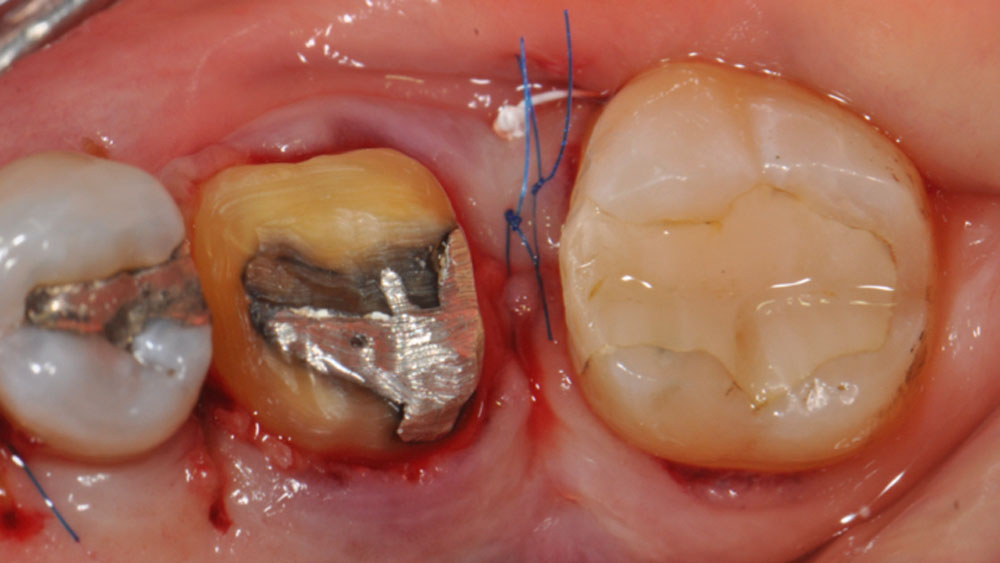
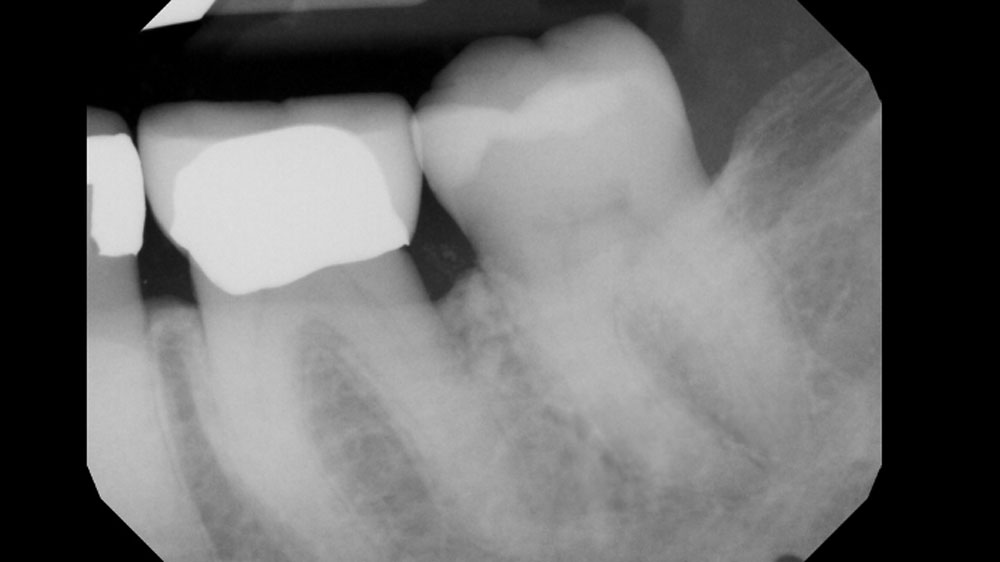
A 68-year-old male patient, who received an implant in tooth position #31 about 8 years prior, presented for an examination. He reports bleeding during brushing around the implant and some discomfort. Clinically, there was severe vertical bone loss, profuse bleeding on probing, and deep probing depths, but no pain. The condition was diagnosed as peri-implantitis according to the 2018 classification.
THE RISK PROFILE
| Low Risk | Medium Risk | High Risk | |
|---|---|---|---|
| Patient’s health | Intact immune system | Light smoker | Impaired immune system |
| Patient’s esthetic requirements | Low | Medium | High |
| Height of smile line | Low | Medium | High |
| Gingival biotype | Thick – “low scalloped” | Medium – “medium scalloped” | Thin – “high scalloped” |
| Infection at implant sight | None | Chronic | Acute |
| Restorative status of adjacent tooth | Intact | Restored | |
| Soft-tissue anatomy | Intact | Compromised | |
| Bone anatomy of the alveolar ridge | No defect | Horizontal defect | Vertical defect |
Additional Risk Factors: The patient exhibited bleeding on probing and deep pocket depths. He also reported occasional marijuana use and was inconsistent with periodontal maintenance and oral hygiene visits.
THE APPROACH
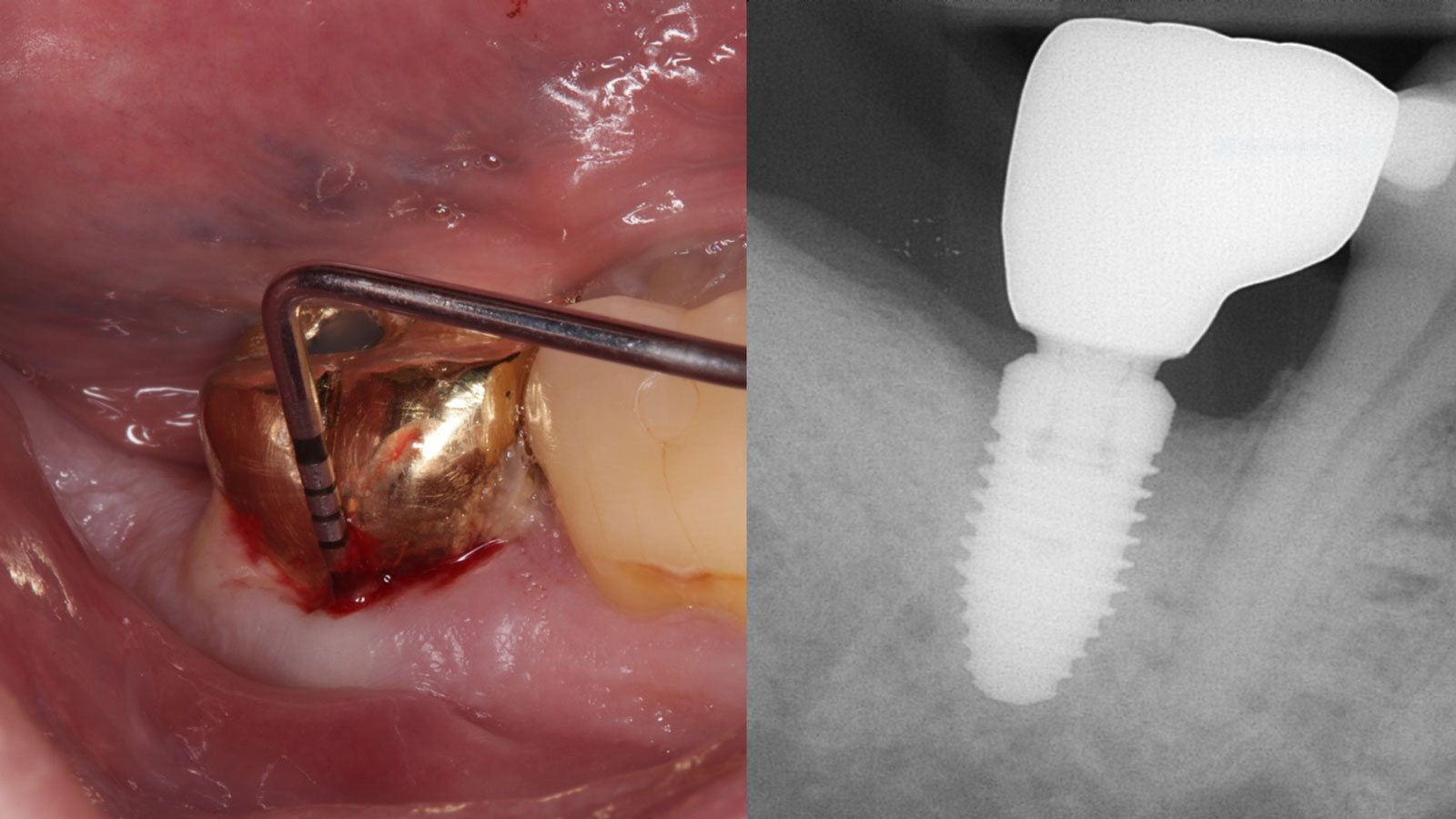
The treatment goals were to eliminate peri-implant infection, regenerate lost hard and soft tissues, and ensure long-term implant stability. A closed regenerative approach was utilized, including crown removal, thorough implant decontamination with Perioflow®, an airpolishing technology, application of the correct bone grafting materials (Geistlich Bio-Oss®, vallos® and GEM 21S®), enclosed healing, and fabrication of a new crown to facilitate hygiene.
“The implant presented with significant bone loss, deep probing depths, and bleeding on probing, placing it at risk of failure and requiring intervention to preserve function and longevity.”
— Andrea Ravidà, DDS, MS, PhD
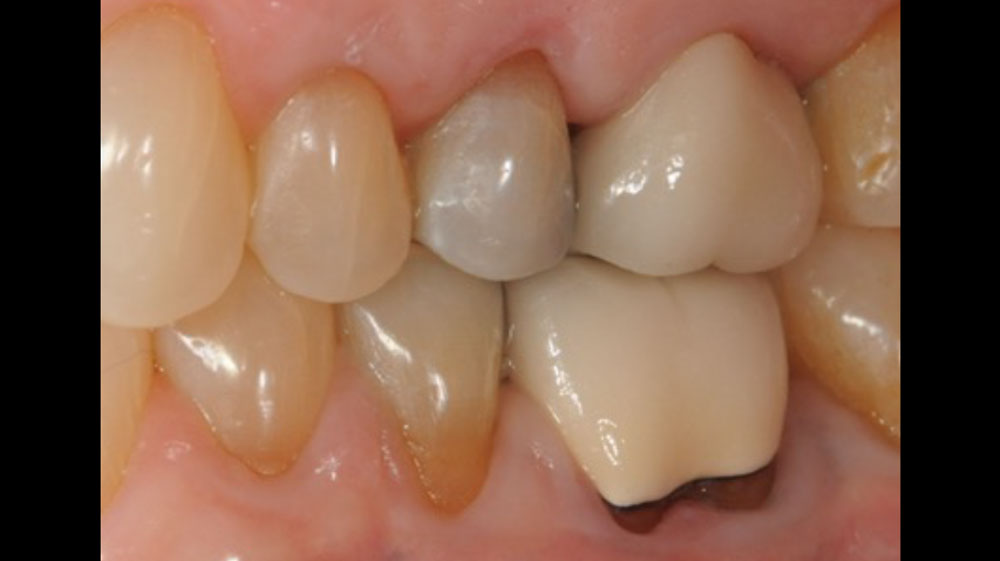
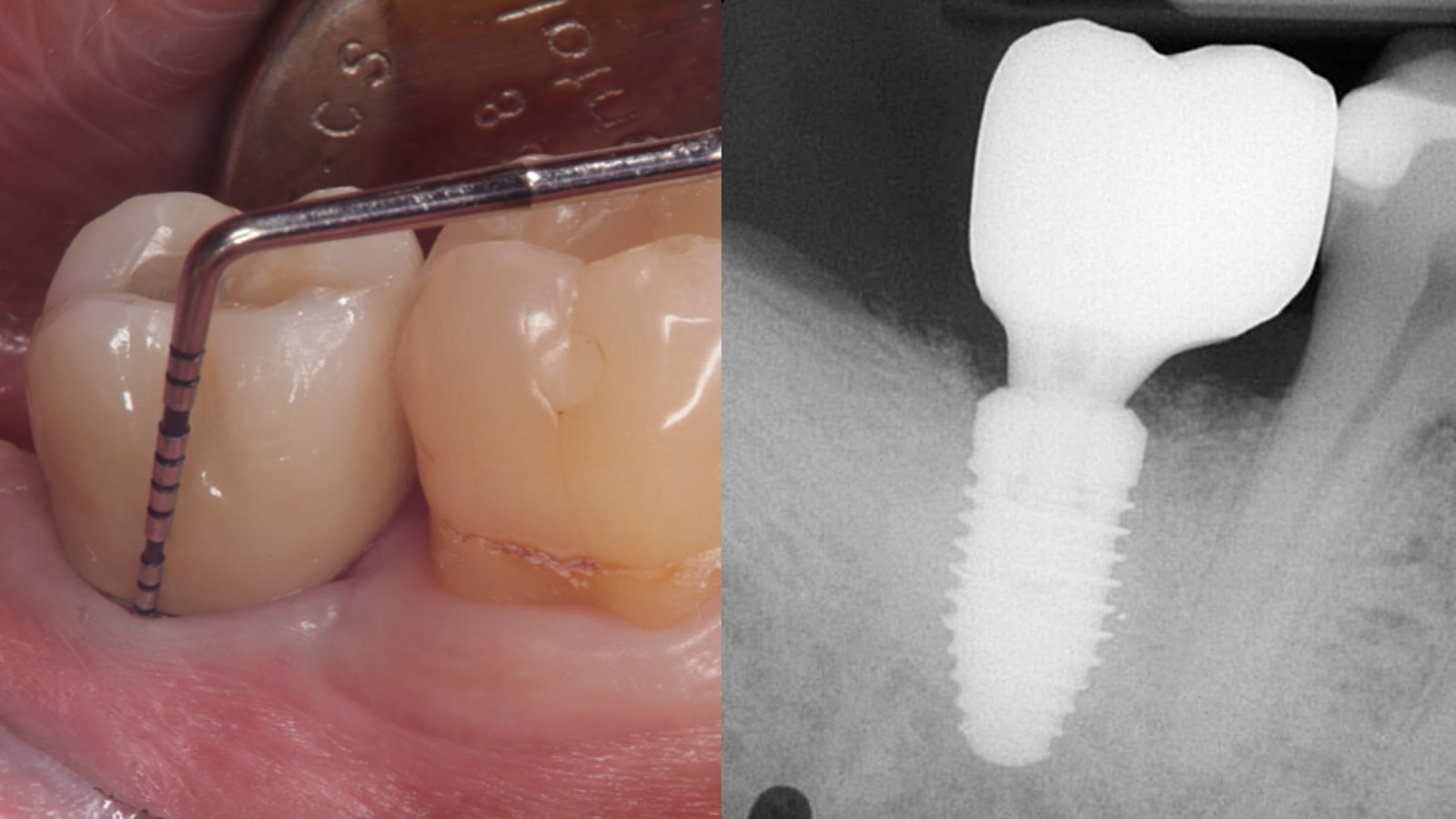
THE OUTCOME
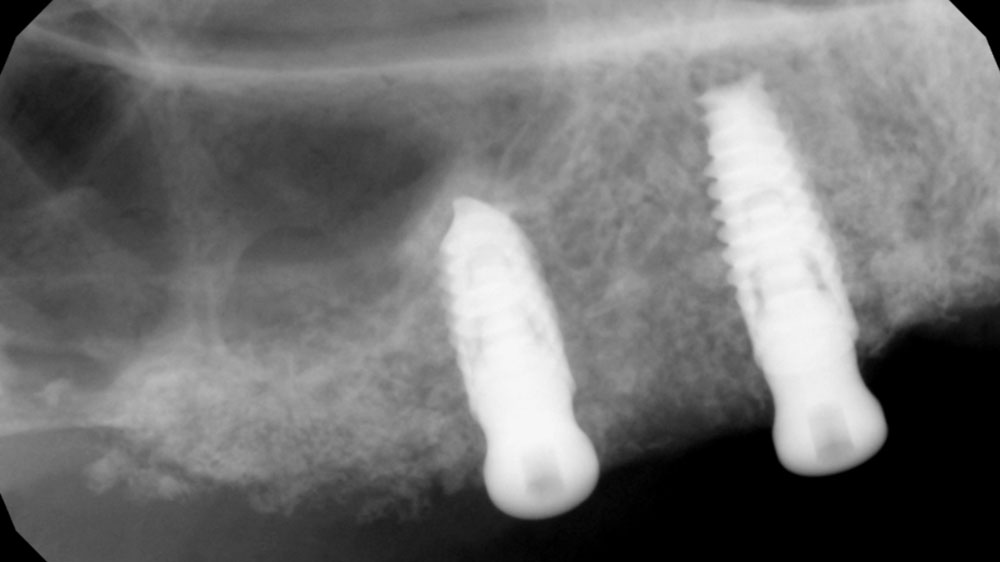
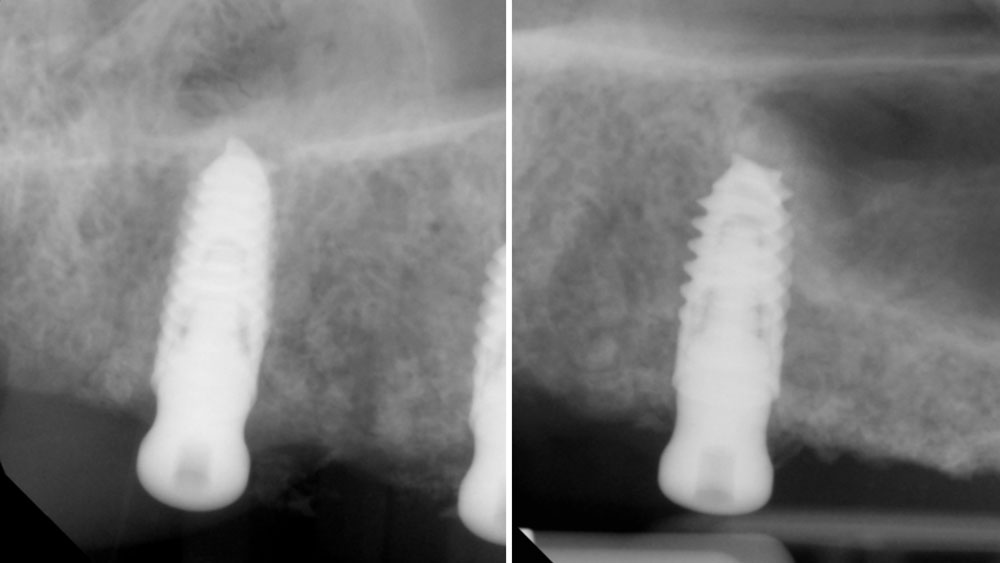
At the two-year follow-up, clinical and radiographic assessments showed disease resolution, complete bone gain, and stable peri-implant tissues. Probing depths were within healthy ranges, and no bleeding on probing was observed, confirming the long-term success of the treatment.

Andrea Ravidà, DDS, MS, PhD
Dr. Andrea Ravidà is the Director of the Graduate Periodontics Program in the department of Periodontics at the University of Pittsburgh. He conducts clinical research focusing on peri-implantitis, periodontitis and short implants. He has published more than 70 peer-reviewed articles and conference abstracts/presentations related to periodontics and implant therapy. He is section editor of the International Journal of Oral Implantology and the Journal of Translational Medicine.

Anu Viswanathan DDS, MDS
Dr. Anu Viswanathan is a Diplomate of the American Board of Periodontology and Implant Dentistry. She earned her Doctor of Dental Surgery degree from the University of Colorado School of Dental Medicine in 2019. Dr. Viswanathan completed a Certificate in Periodontics and earned a Master of Dental Science at the University of Pittsburgh School of Dental Medicine. She also obtained a Certificate in IV Sedation. Dr. Viswanathan is currently in private practice in Shoreline, Connecticut.
Sorry, you do not have permission to view this content.
Sorry, you do not have permission to view this content.
Sorry, you do not have permission to view this content.
Sorry, you do not have permission to view this content.
Sorry, you do not have permission to view this content.
Sorry, you do not have permission to view this content.
Sorry, you do not have permission to view this content.
Sorry, you do not have permission to view this content.
Sorry, you do not have permission to view this content.
Sorry, you do not have permission to view this content.