BIOBRIEF
Vertical Bone Augmentation with Bone Plate Technique and Soft Tissue Phenotype Modification


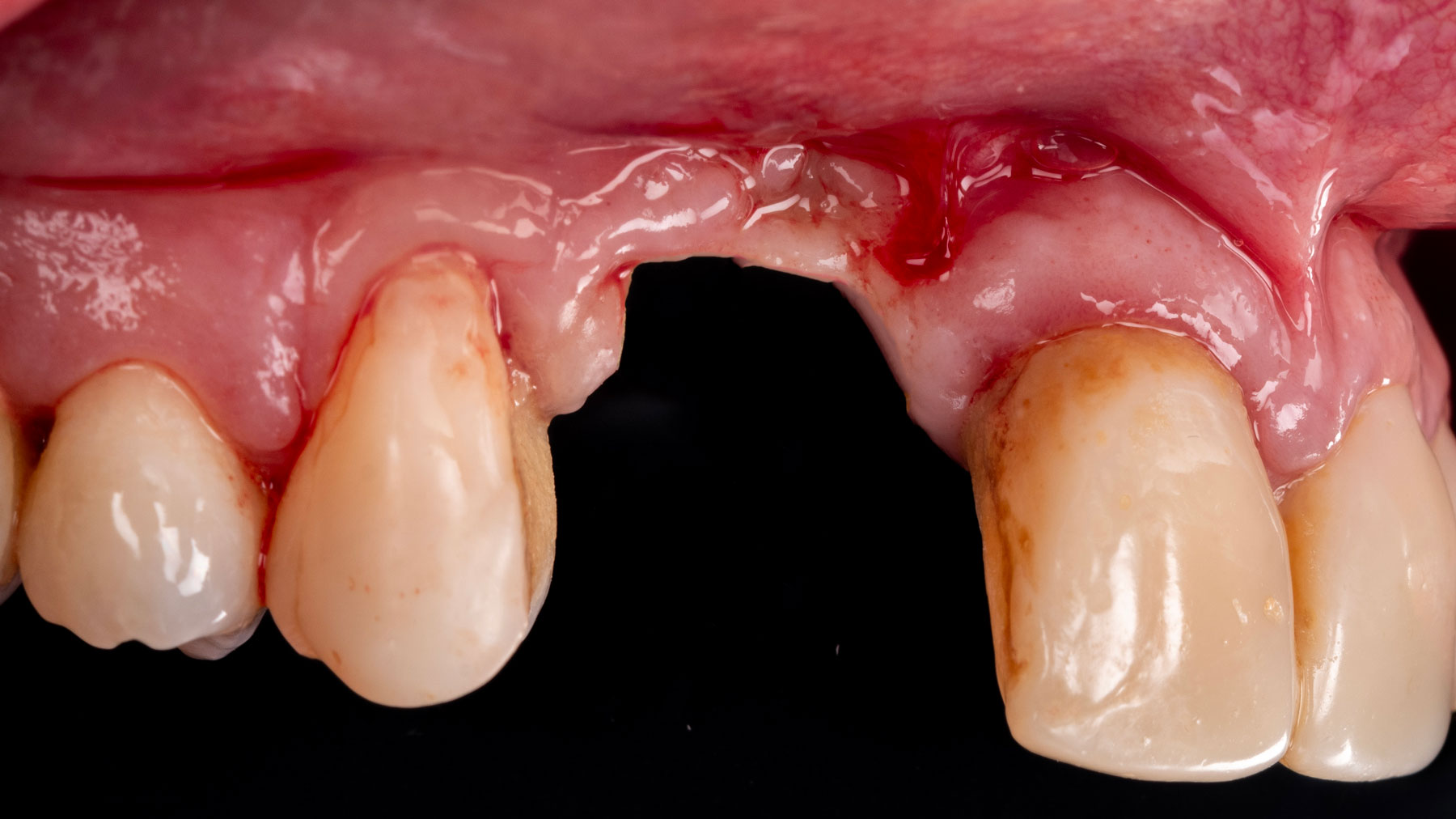
THE RISK PROFILE
| Low Risk | Medium Risk | High Risk | |
|---|---|---|---|
| Patient’s esthetic requirements | Low | Medium | High |
| Height of the smile line | Low | Medium | High |
| Gingival biotype | Thick – “low scalloped” | Medium – “medium scalloped” | Thin – “high scalloped” |
| Infection at implant sight | None | Chronic | Acute |
| Bone height at adjacent tooth | ≤ 5 mm from contact point | 5.5 – 6.5 mm from contact point | ≥ 7 mm from contact point |
| Restorative status of adjacent tooth | Intact | Restored (#8 has been restored) | |
| Width of tooth gap | 1 tooth (≥ 7 mm) | 1 tooth (≤ 7 mm) | 2 teeth or more |
| Soft-tissue anatomy | Intact | Compromised | |
| Bone anatomy of the alveolar ridge | No defect | Horizontal defect | Vertical defect |
THE APPROACH
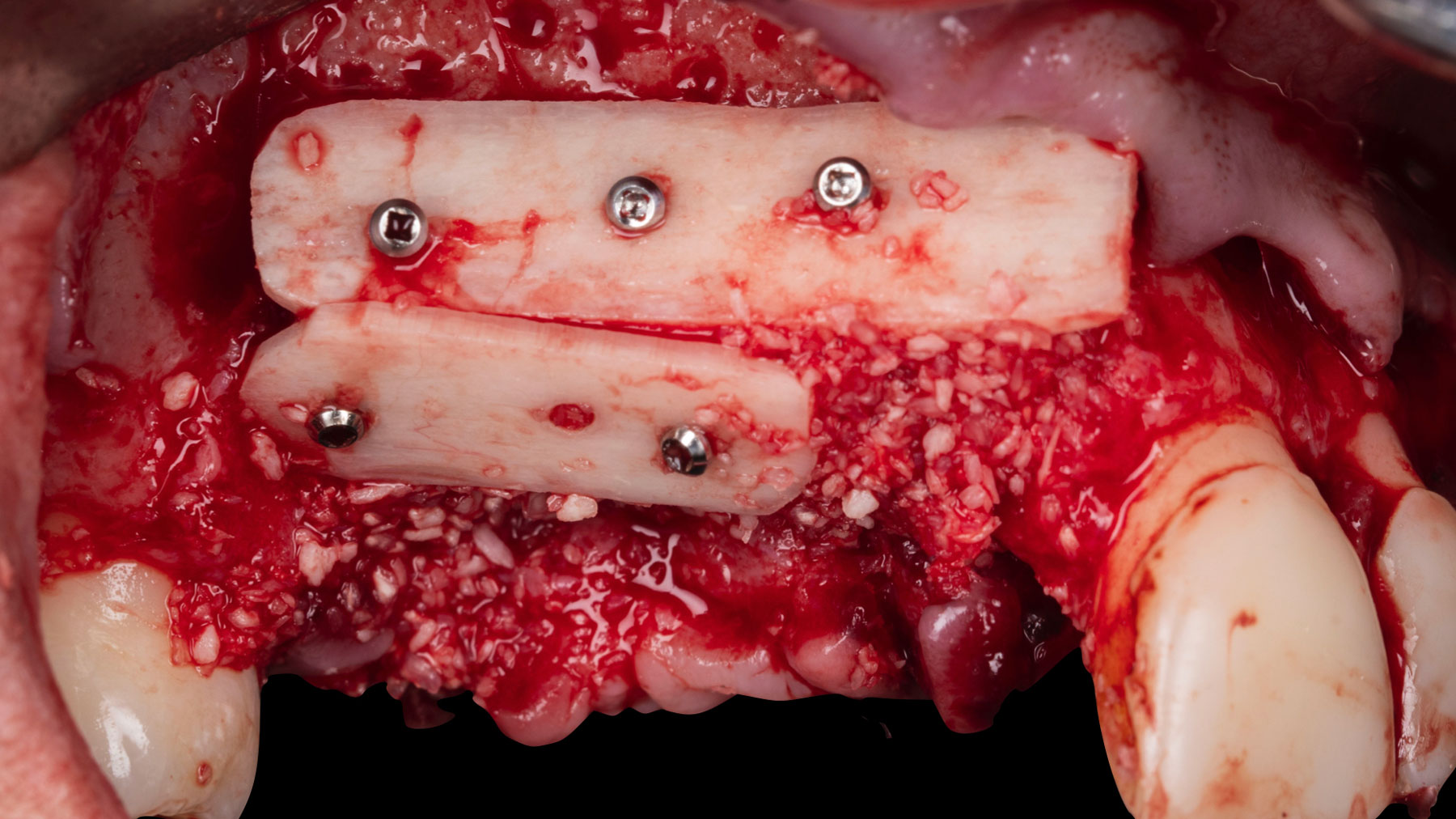
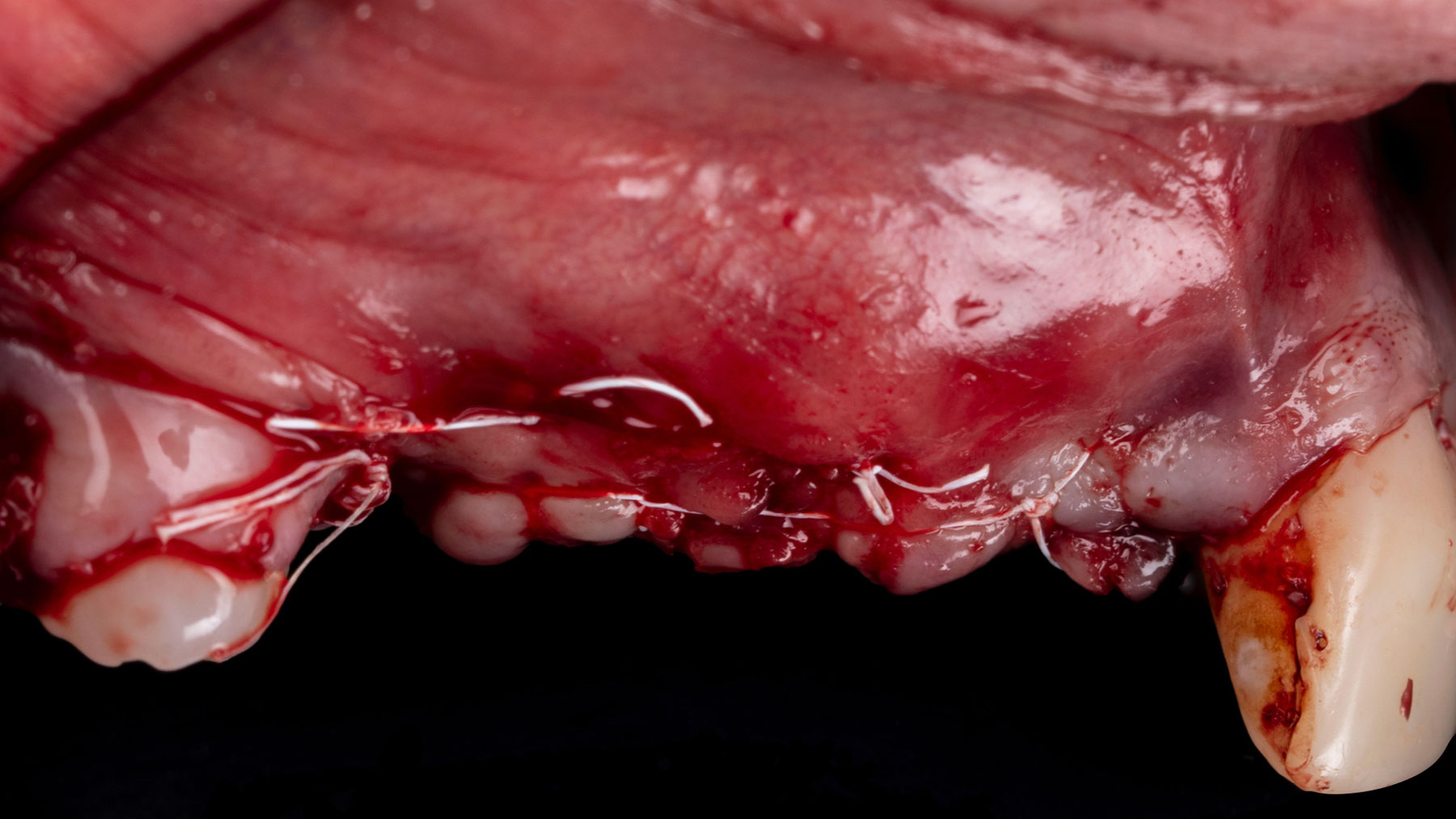
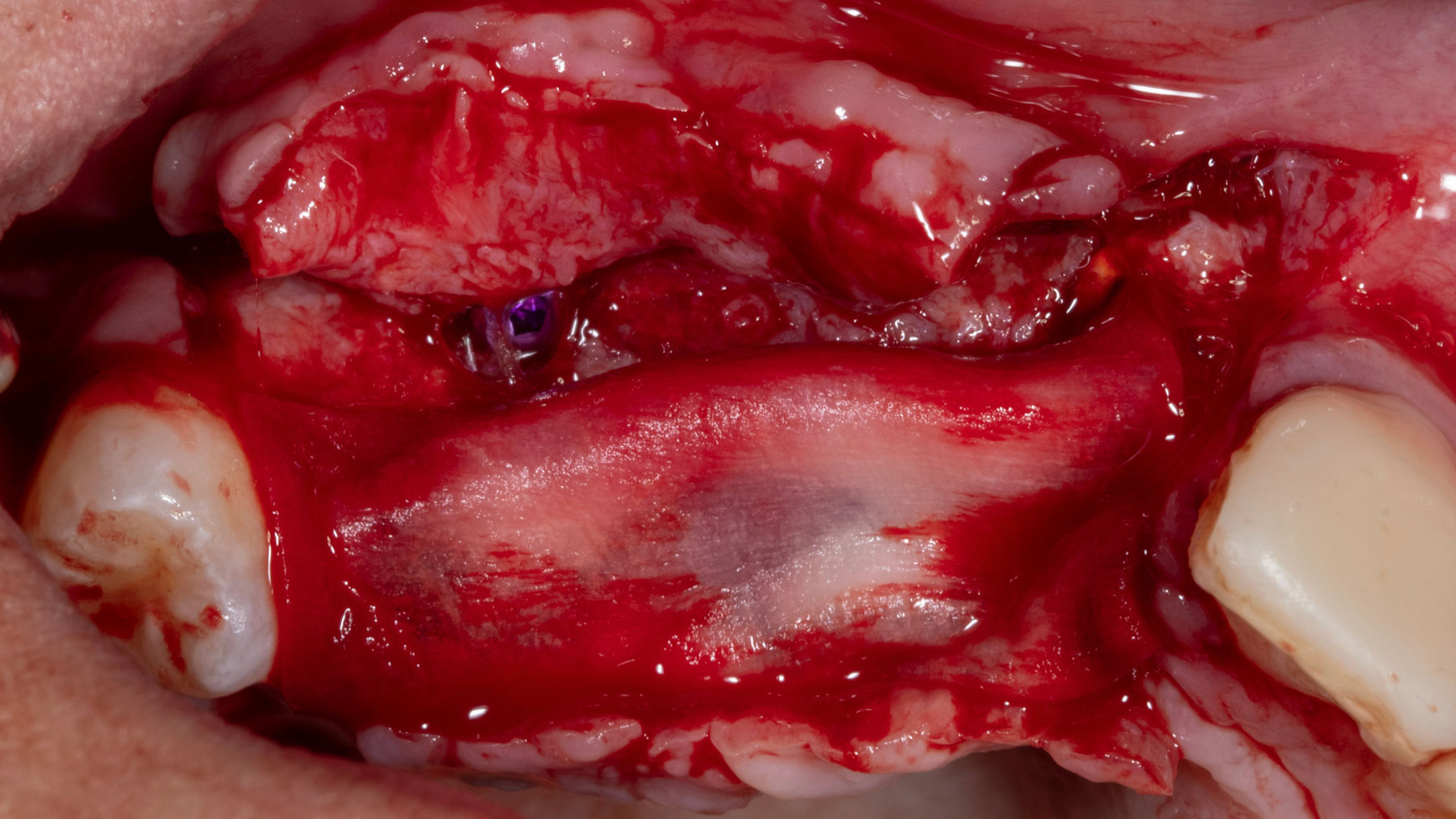
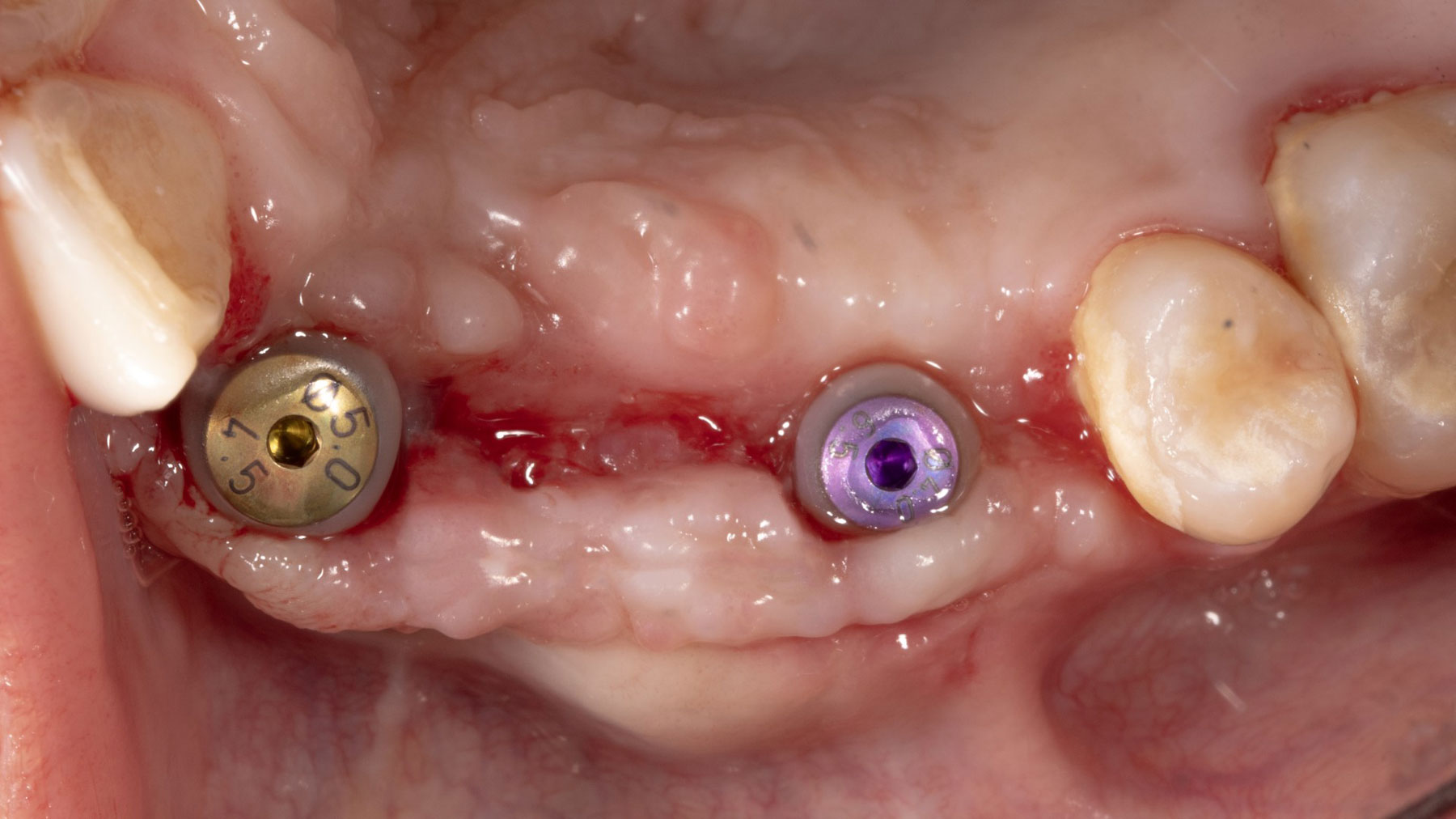
Vertical ridge augmentation was performed using allogenic cortical plates fixed with microscrews to create a stable, space-maintaining box, which was densely packed with vallos® mineralized cortico-cancellous granules hydrated with the rhPDGF-BB component of GEM 21S®. Five months later, the implants were placed, and the peri-implant soft tissue phenotype was enhanced using Geistlich Mucograft® and Geistlich Fibro-Gide® to improve soft-tissue height and thickness.
“The patient had high esthetic demands and specifically wanted to avoid autogenous grafting. Remarkably, this case was completed with 0% autogenous tissue, demonstrating that outcomes traditionally thought to require autogenous grafts can be achieved otherwise.”
— Dr. Muhammad Saleh
THE OUTCOME
Vertical ridge augmentation of the maxillary anterior was achieved using allogenic cortical bone plates fixed with microscrews to create a box configuration, filled with vallos® mineralized cortico-cancellous granules mixed with rhPDGF-BB component of GEM21S®. At implant placement, soft tissue phenotype was enhanced using Geistlich Mucograft® and Geistlich Fibro-Gide®.

Muhammad Saleh, BDS, MSD, PhD
Dr. Muhammad H. Saleh, Diplomate of the American Board of Periodontology, is a full-time faculty member at the University of Michigan and an internationally recognized lecturer in periodontology and implant dentistry. He has published over 140 peer-reviewed articles and reviews for leading Q1 journals. His numerous honors include the 2021 AAPF Schoor Award, 2022 Ramfjord Symposium Award, 2022 AAP Institute Award, 2024 AAPF LEAD and Nevins Awards, and the 2025 Roy H. Roberts Award.