
Find out more about the worldwide leading regeneration company and its gold standard products – Geistlich Bio-Oss® and Geistlich Bio-Gide®.

The No. 1 biomaterials company for regeneration experts.1
For over 30 years, Geistlich has specialized in regenerative biomaterials and has been the world market leader for specialists in regenerative dentistry.
Compliance with international scientific standards has been our core principle since day one. With more than 2,000 studies and scientific publications, Geistlich biomaterials are the most extensively researched products for regenerative dentistry worldwide.2-4
Success in a nutshell

Over 25 million patients treated worldwide5

Every 14 seconds a Geistlich product is used5

Premium Swiss quality

Strictly monitored manufacturing process

Only carefully selected raw materials are used

Highest safety standards and excellent biocompatibility

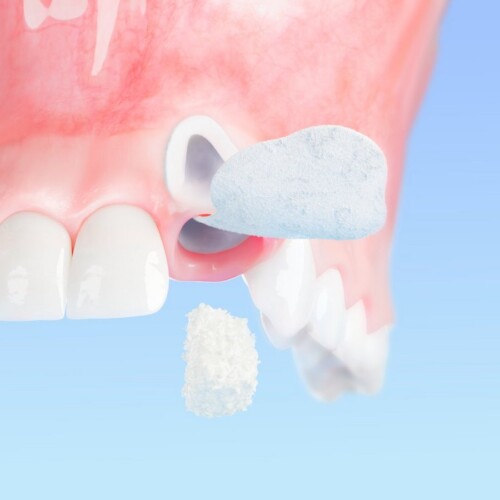
Geistlich Bio-Oss® & Geistlich Bio-Gide®: Versatile solutions, easy handling.
Peace of mind comes with our reliable, easy-to-use products. Geistlich Bio-Oss®, the original bone graft substitute, and Geistlich Bio-Gide®, the original collagen membrane, have been the epitome of predictable and lasting treatment outcomes for decades:
- Bone augmented with Geistlich Bio-Oss® leads to unmatched implant survival rates.6-8
- Geistlich Bio-Gide® ensures reliable bone regeneration and the best possible tissue integration.9-12
Talk to our experts!
Geistlich guarantees consistent, high-quality products delivered on time. Our skilled sales team is always ready to support you, anticipating your needs and providing solutions before you even have to ask.
Interesting facts about Geistlich





represented in over 90 countries.
- iDATA Global Market Report Suite for Dental GL Bone Graft Substitutes and Other Biomaterials, June 2024
- Search conducted on 05.05.2025 in the internal library. Reference list available upon request.
- iData Research Inc., US Dental Bone Graft Substitutes and other Biomaterials Market, 2021.
- iData Research Inc., European Dental Bone Graft Substitutes and other Biomaterials Market, 2021.
- Data on file.
- Jung RE, et al., Clin Oral Implants Res. 2021 Dec;32(12):1455-1465. (clinical study)
- Benic GI, et al., J Clin Periodontol. 2017 Mar;44(3):315-325. (clinical study)
- Chappuis V, et al., J Dent Res. 2018 Mar;97(3):266-274. (clinical study)
- Perelman-Karmon M, et al., Int J Periodontics Restorative Dent. 2012 Aug;32(4):459-65. (clinical study)
- Tal H, et al., Clin Oral Implants Res 2008; 19 (3), 295-302. (clinical study)
- Knöfler W, et al., Int J Implant Dent. 2016 Dec;2(1):25. (clinical study)
- Rothamel D, et al., Clin Oral Implants Res 2005; 16 (3), 369-78. (pre-clinical study)

