Now Available!

Efficient. Essential. Everyday Needs.


Product Information
The Geistlich® Allogenic Bonegraft CSP family offers a complete range of allograft solutions designed to meet everyday regenerative procedure needs.
Each graft is processed using the Cancelle SP® Sterilization Process, a proprietary, validated method that removes debris and inactivates and/or removes a panel of microorganisms while maintaining biocompatibility.1
About Geistlich® Allogenic BoneGraft CSP
- Choice of five configurations for cortical, cancellous, and blended particulates
- Convenient volume options ranging from 0.25 cc to 2.0 cc (varies by type)
- Particle size range: 250–1,000 microns for optimal handling and packing
- 70% mineralized cortical / 30% DBM mix for clinicians seeking both osteoconductive and osteoinductive potential
- Each lot of implants containing DBM is lot release tested for BMP-2.1 BMPs are recognized as a marker of osteoinductive potential.*
* Having the ability to induce bone growth.
Geistlich® Allogenic BoneGraft CSP Configurations

Mineralized Cortical/
Cancellous Natural Blend

Mineralized
Cancellous

Mineralized Cortical

70% Mineralized Cortical/
30% DBM

Cortical Demineralized Bone Matrix
Quality Starts with the Donor

Donor Assessment
Each donor is evaluated for diseases that could impact tissue quality.

Record Review
A comprehensive review of all relevant donor records is conducted prior to tissue release.

Risk Assessment Interview
Independent licensed recovery agencies interview donor’s next of kin to assess donor risk factors.

Physical Assessment
Recovery agency performs a physical examination of the donor to identify any signs of infection or conditions that may adversely affect tissue.
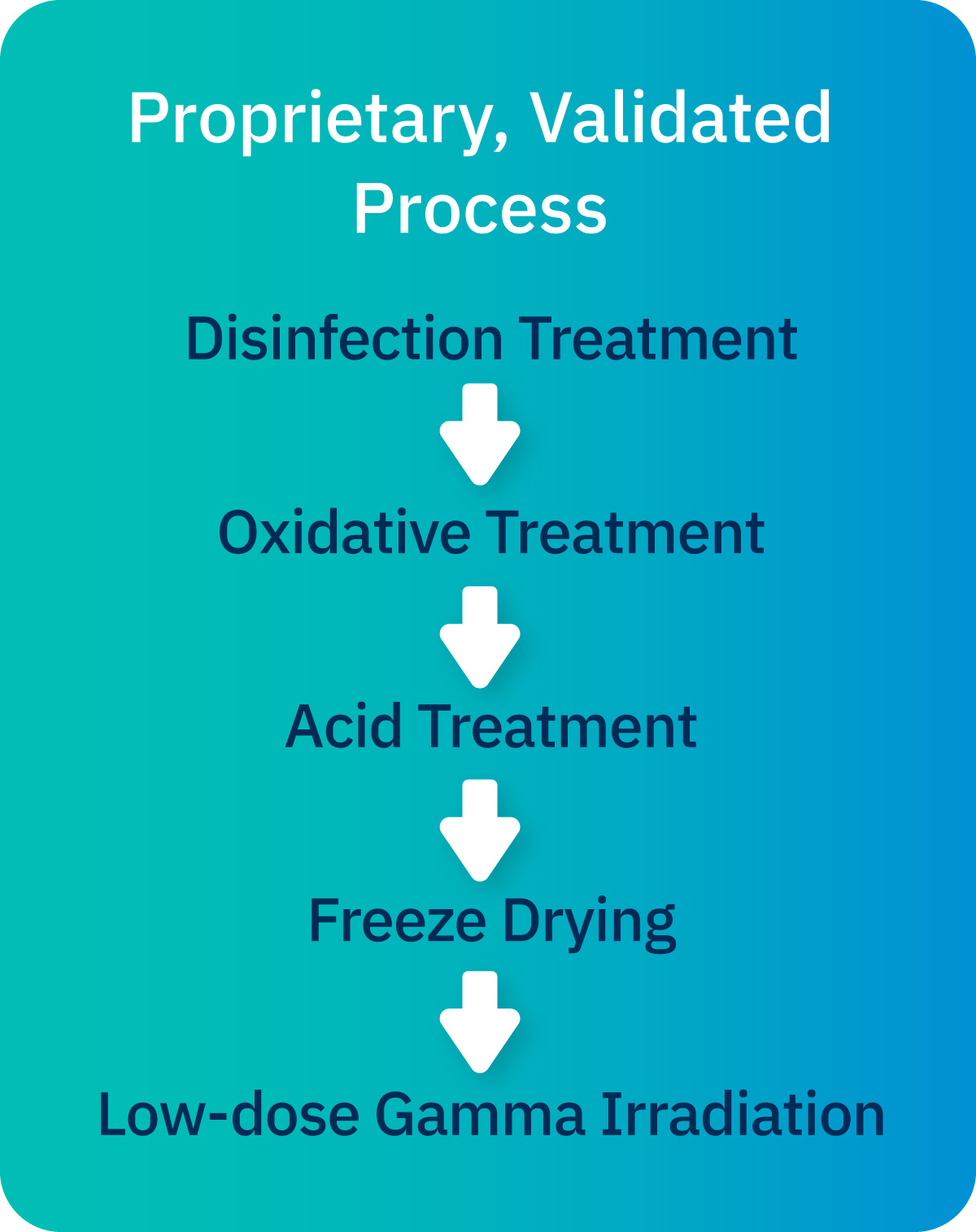
Cancelle SP® Sterilization Process
The Cancelle SP® Sterilization Process is a proprietary process that sterilizes demineralized bone matrix (DBM) and cortical cancellous chips (CCC). This process is validated to inactivate and/or remove a panel of microorganisms without the use of antibiotics.1
- A combination of treatments and washes removes debris from the graft material.
- Cleansing rinses eliminate residual chemicals to ensure biocompatibility.
- Low-temperature, low-dose gamma irradiation helps preserve the utility and biological properties of the graft.
Geistlich® Allogenic
BoneGraft CSP

Frequently Asked Questions
- Data on file with RTI Surgical, Inc.
- Gruskin, E. et al. Demineralized bone matrix in bone repair: History and use. Advanced Drug Delivery Reviews 64 (2012) 1063-1077.
- Data on file with RTI Surgical, Inc.
- Wood and Mealey, Histologic comparison of healing after tooth extraction with ridge preservation using mineralized versus demineralized freeze-dried bone allograft, J Periodontol, March 2012.
- Albrektsson T, Johansson C. Osteoinduction, osteoconduction and osseointegration. Eur Spine J. 2001;10(Suppl 2):S96–S101. doi:10.1007/s005860100282
- Roberts TT, Rosenbaum AJ. Bone grafts, bone substitutes and orthobiologics. Organogenesis. 2012;8(4):114–124. doi:10.4161/org.23306.
- Cancelle SP® is a registered trademark of RTI Surgical, Inc. d/b/a Evergen.
Disclaimer: Lab data may not be representative of effects or performance in humans.



